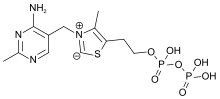
Thiamine
 | |
 Skeletal formula and ball-and-stick model of the thiamine cation | |
| Clinical data | |
|---|---|
| Pronunciation | /ˈθaɪ.əmɪn/ THY-ə-min |
| Other names | Vitamin B1, aneurine, thiamin |
| AHFS/Drugs.com | Monograph |
| License data | |
| Routes of administration | by mouth, IV, IM[1] |
| Drug class | vitamin |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 3.7% to 5.3% (Thiamine hydrochloride)[2] |
| Identifiers | |
| |
JSmol) |
|
| |
| |
Thiamine, also known as thiamin and vitamin B1, is a
Food sources of thiamine include
Thiamine supplements are generally well tolerated.
Definition
Thiamine is one of the
The chemical structure consists of an
Deficiency
Well-known disorders caused by thiamine deficiency include beriberi, Wernicke–Korsakoff syndrome, optic neuropathy, Leigh's disease, African seasonal ataxia (or Nigerian seasonal ataxia), and central pontine myelinolysis.[13] Symptoms include malaise, weight loss, irritability and confusion.[10][14][15]
In Western countries, chronic alcoholism is a risk factor for deficiency. Also at risk are older adults, persons with HIV/AIDS or diabetes, and those who have had bariatric surgery.[1] Varying degrees of thiamine insufficiency have been associated with the long-term use of diuretics.[16][17]
Biological functions

Five natural thiamine phosphate derivatives are known:
Thiamine pyrophosphate
Thiamine pyrophosphate (TPP), also called thiamine diphosphate (ThDP), participates as a coenzyme in metabolic reactions, including those in which
- Present in most species
- α-ketoglutarate dehydrogenase)
- branched-chain α-keto acid dehydrogenase
- 2-hydroxyphytanoyl-CoA lyase
- transketolase
- Present in some species:
- pyruvate decarboxylase (in yeast)
- several additional bacterial enzymes
The enzymes transketolase, pyruvate dehydrogenase (PDH), and 2-oxoglutarate dehydrogenase (OGDH) are important in
Thiamine triphosphate
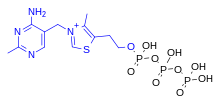
ThTP is implicated in chloride channel activation in the neurons of mammals and other animals, although its role is not well understood.[20] ThTP has been found in bacteria, fungi and plants, suggesting that it has other cellular roles.[23] In Escherichia coli, it is implicated in the response to amino acid starvation.[24]
Adenosine derivatives
AThDP exists in small amounts in vertebrate liver, but its role remains unknown.[24]
AThTP is present in E. coli, where it accumulates as a result of carbon starvation. In this bacterium, AThTP may account for up to 20% of total thiamine. It also exists in lesser amounts in yeast, roots of higher plants and animal tissue.[24]
Medical uses
During pregnancy, thiamine is sent to the fetus via the placenta. Pregnant women have a greater requirement for the vitamin than other adults, especially during the third trimester. Pregnant women with hyperemesis gravidarum are at an increased risk of thiamine deficiency due to losses when vomiting.[25] In lactating women, thiamine is delivered in breast milk even if it results in thiamine deficiency in the mother.[4][26]
Thiamine is important not only for
Dietary recommendations
| US National Academy of Medicine | |
| Age group | RDA (mg/day) |
|---|---|
| Infants 0–6 months | 0.2* |
| Infants 6–12 months | 0.3* |
| 1–3 years | 0.5 |
| 4–8 years | 0.6 |
| 9–13 years | 0.9 |
| Females 14–18 years | 1.0 |
| Males 14+ years | 1.2 |
| Females 19+ years | 1.1 |
| Pregnant/lactating females 14–50 | 1.4 |
| * Adequate intake for infants, as an RDA has yet to be established[4] | |
| European Food Safety Authority | |
| Age group | Adequate intake (mg/MJ)[28] |
| All persons 7 months+ | 0.1 |
| Neither the US National Academy of Medicine nor the European Food Safety Authority have determined the tolerable upper intake level for thiamine[4] | |
The US National Academy of Medicine updated the Estimated Average Requirements (EARs) and Recommended Dietary Allowances (RDAs) for thiamine in 1998. The EARs for thiamine for women and men aged 14 and over are 0.9 mg/day and 1.1 mg/day, respectively; the RDAs are 1.1 and 1.2 mg/day, respectively. RDAs are higher than EARs to provide adequate intake levels for individuals with higher than average requirements. The RDA during pregnancy and for lactating females is 1.4 mg/day. For infants up to the age of 12 months, the Adequate Intake (AI) is 0.2–0.3 mg/day and for children aged 1–13 years the RDA increases with age from 0.5 to 0.9 mg/day.[4]
The
Neither the National Academy of Medicine nor EFSA have set an upper intake level for thiamine, as there is no human data for adverse effects from high doses.[4][28]
Safety
Thiamine is generally well tolerated and non-toxic when administered orally.[7] There are rare reports of adverse side effects when thiamine is given intravenously, including allergic reactions, nausea, lethargy, and impaired coordination.[28][3]
Labeling
For US food and dietary supplement labeling purposes, the amount in a serving is expressed as a percent of Daily Value. Since May 27, 2016, the Daily Value has been 1.2 mg, in line with the RDA.[30][31]
Sources
Thiamine is found in a wide variety of processed and whole foods,
Food fortification
Some countries require or recommend fortification of grain foods such as
Synthesis
Biosynthesis
Thiamine biosynthesis occurs in bacteria, some protozoans, plants, and fungi.[37][38] The thiazole and pyrimidine moieties are biosynthesized separately and are then combined to form ThMP by the action of thiamine-phosphate synthase.
The pyrimidine ring system is formed in a reaction catalysed by
The starting material is
The thiazole ring is formed in a reaction catalysed by thiazole synthase (EC 2.8.1.10).[39] The ultimate precursors are 1-deoxy-D-xylulose 5-phosphate, 2-iminoacetate and a sulfur carrier protein called ThiS. An additional protein, ThiG, is also required to bring together all the components of the ring at the enzyme active site.[43]

The final step to form ThMP involves decarboxylation of the thiazole intermediate, which reacts with the pyrophosphate derivative of phosphomethylpyrimidine, itself a product of a kinase, phosphomethylpyrimidine kinase.[39]
The biosynthetic pathways differ among organisms. In E. coli and other enterobacteriaceae, ThMP is phosphorylated to the cofactor TPP by a thiamine-phosphate kinase (ThMP + ATP → TPP + ADP).[39] In most bacteria and in eukaryotes, ThMP is hydrolyzed to thiamine and then pyrophosphorylated to TPP by thiamine diphosphokinase (thiamine + ATP → TPP + AMP).[44]
The biosynthetic pathways are regulated by
Laboratory synthesis
In the first
Industrial synthesis

Synthetic analogues
Many vitamin B1 analogues, such as Benfotiamine, fursultiamine, and sulbutiamine, are synthetic derivatives of thiamine. Most were developed in Japan in the 1950s and 1960s as forms that were intended to improve absorption compared to thiamine.[51] Some are approved for use in some countries as a drug or non-prescription dietary supplement for treatment of diabetic neuropathy or other health conditions.[52][53][54]
Absorption, metabolism and excretion
In the upper small intestine, thiamine phosphate esters present in food are hydrolyzed by alkaline
The majority of thiamine in
Human storage of thiamine is about 25 to 30 mg, with the greatest concentrations in skeletal muscle, heart, brain, liver, and kidneys. ThMP and free (unphosphorylated) thiamine are present in plasma, milk, cerebrospinal fluid, and, it is presumed, all extracellular fluid. Unlike the highly phosphorylated forms of thiamine, ThMP and free thiamine are capable of crossing cell membranes. Calcium and magnesium have been shown to affect the distribution of thiamine in the body and magnesium deficiency has been shown to aggravate thiamine deficiency.[20] Thiamine contents in human tissues are less than those of other species.[14][55]
Thiamine and its metabolites (2-methyl-4-amino-5-pyrimidine carboxylic acid, 4-methyl-thiazole-5-acetic acid, and others) are excreted principally in the urine.[3]
Interference
The bioavailability of thiamine in foods can be interfered with in a variety of ways. Sulfites, added to foods as a preservative,[56] will attack thiamine at the methylene bridge, cleaving the pyrimidine ring from the thiazole ring. The rate of this reaction is increased under acidic conditions.[14] Thiamine is degraded by thermolabile thiaminases present in some species of fish, shellfish and other foods.[10] The pupae of an African silk worm, Anaphe venata, is a traditional food in Nigeria. Consumption leads to thiamine deficiency.[57] Older literature reported that in Thailand, consumption of fermented, uncooked fish caused thiamine deficiency, but either abstaining from eating the fish or heating it first reversed the deficiency.[58] In ruminants, intestinal bacteria synthesize thiamine and thiaminases. The bacterial thiaminases are cell surface enzymes that must dissociate from the cell membrane before being activated; the dissociation can occur in ruminants under acidotic conditions. In dairy cows, over-feeding with grain causes subacute ruminal acidosis and increased ruminal bacteria thiaminase release, resulting in thiamine deficiency.[59]
From reports on two small studies conducted in Thailand, chewing slices of
Bariatric surgery for weight loss is known to interfere with vitamin absorption.[61] A meta-analysis reported that 27% of people who underwent bariatric surgeries experience vitamin B1 deficiency.[62]
History
Thiamine was the first of the water-soluble vitamins to be isolated.[63] The earliest observations in humans and in chickens had shown that diets of primarily polished white rice caused beriberi, but did not attribute it to the absence of a previously unknown essential nutrient.[64][65]
In 1884,
The specific connection to grain was made in 1897 by
In 1910, a Japanese agricultural chemist of
Sir
- Some contributors to the discovery of thiamine
References
- ^ a b c d e f "Thiamin Fact Sheets for Health Professionals". Office of Dietary Supplements. 11 February 2016. Archived from the original on 30 December 2016. Retrieved 30 December 2016.
- PMID 22305197.
- ^ ISBN 978-0-323-66162-1.
- ^ ISBN 978-0-309-06554-2. Archivedfrom the original on 16 July 2015. Retrieved 29 August 2017.
- ^ "Thiamine: MedlinePlus Drug Information". medlineplus.gov. Archived from the original on 28 April 2018. Retrieved 30 April 2018.
- ^ a b "Thiamin". Micronutrient Information Center, Linus Pauling Institute, Oregon State University. 2013. Archived from the original on 2 February 2017. Retrieved 2 February 2022.
- ^ a b c d American Society of Health-System Pharmacists. "Thiamine Hydrochloride". Drugsite Trust (Drugs.com). Archived from the original on 9 August 2020. Retrieved 17 April 2018.
- ISBN 9781455775668.
There are no cases of adverse effects of excess thiamine... A few isolated cases of puritis...
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ ISBN 978-0-7216-7904-4.
- ^ a b c Butterworth RF (2006). "Thiamin". In Shils ME, Shike M, Ross AC, Caballero B, Cousins RJ (eds.). Modern Nutrition in Health and Disease (10th ed.). Baltimore: Lippincott Williams & Wilkins.
- ISBN 9780123786319.
- ISBN 978-1-60761-310-7.
- ^ ISBN 978-0-12-183493-7.
- PMID 33305487.
- PMID 26899752.
- PMID 33576090.
- ^ PMID 32554808.
- PMID 26212886.
- ^ PMID 16550223.
- from the original on 1 December 2022. Retrieved 1 December 2022.
- PMID 12651851.
- S2CID 25400487.
- ^ PMID 34827643.
- S2CID 84184482.
- PMID 11722950.
- PMID 28915355.
- ^ a b c Tolerable Upper Intake Levels For Vitamins And Minerals (PDF), European Food Safety Authority, 2006, archived (PDF) from the original on 16 March 2016
- ^ "Overview on Dietary Reference Values for the EU population as derived by the EFSA Panel on Dietetic Products, Nutrition and Allergies" (PDF). 2017. Archived (PDF) from the original on 28 August 2017.
- ^ "Federal Register May 27, 2016 Food Labeling: Revision of the Nutrition and Supplement Facts Labels. FR page 33982" (PDF). Archived (PDF) from the original on 8 August 2016.
- ^ "Daily Value Reference of the Dietary Supplement Label Database (DSLD)". Dietary Supplement Label Database (DSLD). Archived from the original on 7 April 2020. Retrieved 6 February 2022.
- ^ "Thiamin content per 100 grams; select food subset, abridged list by food groups". United States Department of Agriculture, Agricultural Research Service, USDA Branded Food Products Database v.3.6.4.1. 17 January 2017. Archived from the original on 2 February 2017. Retrieved 27 January 2017.
- PMID 27745590.
- ^ "What nutrients are added to flour and rice in fortification?". Food Fortification Initiative. 2021. Archived from the original on 8 October 2021. Retrieved 8 October 2021.
- ^ "Map: Count of Nutrients In Fortification Standards". Global Fortification Data Exchange. Archived from the original on 11 April 2019. Retrieved 11 October 2021.
- ^ "Direction under Section 16(5) of Foods Safety and Standards Act, 2006 regarding Operationalisation of Food Safety & Standards (Fortification of Foods) Regulations, 2017 relating to standards for fortification of food" (PDF). Food Safety & Standards Authority of India (FSSAI). 19 May 2017. Archived (PDF) from the original on 17 December 2021. Retrieved 1 February 2022.
- PMID 17898894.
- PMID 18314013.
- ^ a b c d Caspi R (14 September 2011). "Pathway: superpathway of thiamine diphosphate biosynthesis I". MetaCyc Metabolic Pathway Database. Archived from the original on 1 February 2022. Retrieved 1 February 2022.
- PMID 30097089.
- PMID 20886485.
- PMID 26237670.
- PMID 16453030.
- ^ Caspi R (23 September 2011). "Pathway: superpathway of thiamine diphosphate biosynthesis III (eukaryotes)". MetaCyc Metabolic Pathway Database. Archived from the original on 14 November 2022. Retrieved 14 November 2022.
- PMID 18778966.
- PMID 29208764.
- ^ PMID 23208776.
- .
- S2CID 242772232.
- ^ "Substance Infocard". echa.europa.eu. Archived from the original on 20 April 2021. Retrieved 11 May 2022.
- ISBN 9781466515574.
- PMID 34040901.
- ISBN 9780813804286.
- from the original on 25 September 2012. Retrieved 17 July 2022.
- S2CID 7161882.
- ^ McGuire M, Beerman KA (2007). Nutritional Sciences: From Fundamentals to Foods. California: Thomas Wadsworth.
- PMID 10827220.
- ^ PMID 803009.
- S2CID 51606809.
- S2CID 40854060.
- S2CID 245655046.
- S2CID 250149680.
- ^ from the original on 21 June 2020. Retrieved 2 May 2018.
- ^ Houghton Mifflin.
- ^ from the original on 9 August 2020. Retrieved 4 July 2019.
- ^ "The Nobel Prize and the Discovery of Vitamins". nobelprize.org. Archived from the original on 16 January 2018. Retrieved 1 May 2018.
- ^ Grijns G (1901). "Over polyneuritis gallinarum" [On polyneuritis gallinarum]. Geneeskundig Tijdschrift voor Nederlandsch-Indië (Medical Journal for the Dutch East Indies). 41 (1): 3–11. Archived from the original on 29 August 2021. Retrieved 5 February 2020.
- PMID 16993097.
- ^ Funk C (1912). "The etiology of the deficiency diseases. Beri-beri, polyneuritis in birds, epidemic dropsy, scurvy, experimental scurvy in animals, infantile scurvy, ship beri-beri, pellagra". Journal of State Medicine. 20: 341–68. Archived from the original on 4 July 2020. Retrieved 5 February 2020. The word "vitamine" is coined on p. 342: "It is now known that all these diseases, with the exception of pellagra, can be prevented and cured by the addition of certain preventative substances; the deficient substances, which are of the nature of organic bases, we will call "vitamines"; and we will speak of a beri-beri or scurvy vitamine, which means a substance preventing the special disease."
- ^ Jansen BC, Donath WF (1926). "On the isolation of antiberiberi vitamin". Proc. Kon. Ned. Akad. Wet. 29: 1390–400.
- .
- .
- ^ Lohmann K, Schuster P (1937). "Untersuchungen über die Cocarboxylase". Biochem. Z. 294: 188–214.
External links
- "Thiamine". Drug Information Portal. US National Library of Medicine.