Pernicious anemia
| Pernicious anemia | |
|---|---|
| Other names | Vitamin B12 deficiency anemia, Biermer's anemia, bone marrow tests[6] |
| Treatment | Vitamin B12 injections[7] |
| Prognosis | With treatment a normal life[5] |
| Frequency | 1 per 1000 people[8] |
Pernicious anemia is a disease where not enough
Pernicious anemia refers to a type of
Because pernicious anemia is due to a lack of intrinsic factor, it is not preventable.[11] Pernicious anemia can be treated with injections of vitamin B12.[7] If the symptoms are serious, frequent injections are typically recommended initially.[7] There are not enough studies that pills are effective in improving or eliminating symptoms.[12] Often, treatment may be needed for life.[13]
Pernicious anemia due to autoimmune problems occurs in about one per 1000 people in the US. Among those over the age of 60, about 2% have the condition.[8] It more commonly affects people of northern European descent.[2] Women are more commonly affected than men.[14] With proper treatment, most people live normal lives.[5] Due to a higher risk of stomach cancer, those with pernicious anemia should be checked regularly for this.[13] The first clear description was by Thomas Addison in 1849.[15][16] The term "pernicious" means "deadly", and this term came into use because, before the availability of treatment, the disease was often fatal.[5][17]
Signs and symptoms
Pernicious anemia often presents slowly, and can cause harm insidiously and unnoticeably. Untreated, it can lead to neurological complications, and in serious cases, death.
In more severe or prolonged cases of pernicious anemia, nerve cell damage may occur. This is may result in sense loss, difficulty in
Causes
Vitamin B12 cannot be produced by the human body, and must be obtained from the diet. When foods containing B12 are eaten, the vitamin is usually bound to protein and is released by proteases released by the pancreas into the small bowel. Following its release, most B12 is absorbed by the body in the small bowel (

PA may be considered as an end stage of autoimmune
Although the exact role of
Impaired B12 absorption can also occur following gastric removal (gastrectomy) or gastric bypass surgery. In these surgeries, either the parts of the stomach that produce gastric secretions are removed or they are bypassed. This means intrinsic factor, as well as other factors required for B12 absorption, are not available. However, B12 deficiency after gastric surgery does not usually become a clinical issue. This is probably because the body stores many years' worth of B12 in the liver and gastric surgery patients are adequately supplemented with the vitamin.[43][44]
Although no specific PA susceptibility genes have been identified, a genetic factor likely is involved in the disease. Pernicious anemia is often found in conjunction with other autoimmune disorders, suggesting common autoimmune susceptibility genes may be a causative factor.[35] In spite of that, previous family studies and case reports focusing on PA have suggested that there is a tendency of genetic heritance of PA in particular, and close relatives of the PA patients seem to have higher incidence of PA and associated PA conditions.[45][46][47] Moreover, it was further indicated that the formation of antibodies to gastric cells was autosomal dominant gene determined, and the presence of antibodies to the gastric cells might not be necessarily related to the occurrence of atrophic gastritis related to PA.[45][47]
Pathophysiology
Although the healthy body stores three to five years' worth of B12 in the liver, the usually undetected autoimmune activity in one's gut over a prolonged period of time leads to B12 depletion and the resulting anemia; pernicious anemia refers to one of the hematologic manifestations of chronic auto-immune gastritis, in which the immune system targets the parietal cells of the stomach or intrinsic factor itself, leading to decreased absorption of vitamin B12. The body needs enough intrinsic factor to absorb and reabsorb vitamin B12 from the bile, in which reduces the time needed to develop a deficiency.[48]
B12 is required by enzymes for two reactions: the conversion of
Diagnosis

The insidious nature of PA may mean that diagnosis is delayed.
The presence of antibodies to gastric parietal cells and intrinsic factor is common in PA. Parietal cell antibodies are found in other autoimmune disorders and also in up to 10% of healthy individuals. However, around 85% of PA patients have parietal cell antibodies, which means they are a sensitive marker for the disease. Intrinsic factor antibodies are much less sensitive than parietal cell antibodies, but they are much more specific. They are found in about half of PA patients and are very rarely found in other disorders. These antibody tests can distinguish between PA and food-B12 malabsorption.[56][57]
A buildup of certain metabolites occurs in B12 deficiency due to its role in metabolic processes and cellular functions. Methylmalonic acid (MMA) can be measured in both the blood and urine, whereas homocysteine is only measured in the blood. An increase in both MMA and homocysteine distinguishes B12deficiency from folate deficiency because homocysteine alone increases in the latter.[56][57]
Elevated gastrin levels can be found in around 80–90% of PA cases, but they may also be found in other forms of gastritis. Decreased pepsinogen I levels or a decreased pepsinogen I to pepsinogen II ratio may also be found, although these findings are less specific to PA and can be found in food-B12 malabsorption and other forms of gastritis.[57]
The diagnosis of atrophic gastritis type A should be confirmed by gastroscopy and stepwise biopsy.[58] About 90% of individuals with PA have antibodies for parietal cells; however, only 50% of all individuals in the general population with these antibodies have pernicious anemia.[59]
Differential diagnosis
Forms of vitamin B12 deficiency other than PA must be considered in the
The classic test for PA, the
Vitamin B12 deficiency is also prevalent in patients having Crohn's disease (CD) so it should be differentiated.[61]
Treatment
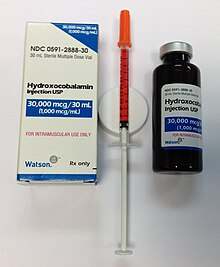
Pernicious anemia is usually easily treated by providing the necessary level of vitamin B12 supplementation.[62] Pernicious anemia can be treated with intramuscular injections of vitamin B12.[63] Initially in high daily doses, followed by less frequent lower doses, as the condition improves.[63] Activity may need to be limited during the course of treatment.[7] As long as the body is saturated with vitamin B12 expected to result in cessation of anemia-related symptoms and there are no other symptoms, unless there are irreversible neurological complications.[62] There are not enough studies on whether pills are as effective in improving or eliminating symptoms as parenteral treatment.[12] Folate supplementation may affect the course and treatment of pernicious anemia if vitamin B12 not replaced.[64] In some severe cases of anemia, a blood transfusion may be needed to resolve haematological effects.[5] Treatment is lifelong.[5][65]
The treatment of PA varies by country and area. Opinions vary over the efficacy of administration (parenteral/oral), the amount and time interval of the doses, or the forms of vitamin B12 (e.g. cyanocobalamin/hydroxocobalamin). More comprehensive studies are still needed in order to validate the feasibility of a particular therapeutic method for PA in clinical practices.[citation needed]
Prognosis
A person with well-treated PA can live a healthy life. Failure to diagnose and treat in time, however, may result in permanent neurological damage, excessive fatigue, depression, memory loss, and other complications. In severe cases, the neurological complications of pernicious anemia can lead to death – hence the name, "pernicious", meaning deadly.[citation needed]
There is an increased risk of
Epidemiology
PA is estimated to affect 0.1% of the general population and 1.9% of those over 60, accounting for 20–50% of B12 deficiency in adults.[1] A review of literature shows that the prevalence of PA is higher in Northern Europe, especially in Scandinavian countries, and among people of African descent, and that increased awareness of the disease and better diagnostic tools might play a role in apparently higher rates of incidence.[68]
History
A case of anemia with a first recognition of associated atrophic gastritis a feature of pernicious anemia, was first described in 1824 by James Combe. This was fully investigated in 1849, by British physician Thomas Addison, from which it acquired the common name of Addison's anemia. In 1871, the first accurate description of the disease in continental Europe was made by Michael Anton Biermer, a German physician who noted the insidious course of the condition. Because it was untreatable and fatal at the time, he first referred to it as "pernicious" anemia.[69][70] Russell coined the term subacute combined degeneration of spinal cord.[71]
In 1907,

Around 1926,
It is not easy to eat uncooked liver, and extracts were developed as a concentrate of liver juice for
Understanding of the pathogenesis of pernicious anaemia increased over subsequent decades. It had long been known that the disease was associated with
A medical author takes the view that Mary Todd Lincoln, the wife of American President Abraham Lincoln, had pernicious anemia for decades and died from it in 1882.[76][77]
Research
Permeation enhancers
Treatment using oral drugs is an easier option in management but the bioavailabity of B12 is low. This is due to low absorption in the intestine, and breakdown by enzyme activity. Research continues to focus on the use of permeation enhancers or permeation absorbers in combination with the treatment. One of the better performing enhancers studied is salcoprozate sodium (SNAC).[78][79] SNAC is able to form a noncovalent complex with cobalamin while preserving its chemical integrity and protect B12 from gastric acidity.[80] This complex is much more lipophilic than the water-soluble vitamin B12, so is able to pass through cellular membranes with greater ease. Molecular dynamics are used in experiments to gain an understanding of the molecular interactions involved in the different molecules used and the degree of ease achieved in absorption across the gastric epithelium.[78]
References
- ^ PMID 23028239.
- ^ ISBN 978-0-7817-7899-2.
- ISBN 978-0-7817-5443-9.
- ^ a b c "What Are the Signs and Symptoms of Pernicious Anemia?". National Heart, Lung, and Blood Institute. April 1, 2011. Archived from the original on 14 March 2016. Retrieved 14 March 2016.
- ^ a b c d e f g h i j k "What Is Pernicious Anemia?". National Heart, Lung, and Blood Institute. April 1, 2011. Archived from the original on 14 March 2016. Retrieved 14 March 2016.
- ^ a b c "How Is Pernicious Anemia Diagnosed?". National Heart, Lung, and Blood Institute. April 1, 2011. Archived from the original on 14 March 2016. Retrieved 14 March 2016.
- ^ a b c d "How Is Pernicious Anemia Treated?". National Heart, Lung, and Blood Institute. April 1, 2011. Archived from the original on 14 March 2016. Retrieved 14 March 2016.
- ^ PMID 33505716.
- ^ a b "What Causes Pernicious Anemia?". National Heart, Lung, and Blood Institute. April 1, 2011. Archived from the original on 14 March 2016. Retrieved 14 March 2016.
- ^ PMID 32053765.
- ^ "How Can Pernicious Anemia Be Prevented?". National Heart, Lung, and Blood Institute. April 1, 2011. Archived from the original on 14 March 2016. Retrieved 14 March 2016.
- ^ PMID 31193945.
- ^ a b "Living With Pernicious Anemia". National Heart, Lung, and Blood Institute. April 1, 2011. Archived from the original on 14 March 2016. Retrieved 14 March 2016.
- ISBN 978-0-7817-6507-7. Archivedfrom the original on 2016-03-14.
- ISBN 978-0-8018-7029-3. Archivedfrom the original on 2016-03-14.
- ISBN 978-1-135-92841-4. Archivedfrom the original on 2016-03-14.
- ISBN 978-0-8036-5791-5. Archivedfrom the original on 2016-03-17.
- ^ "Pernicious Anemia Symptoms, Types, Causes & Treatment". MedicineNet. Retrieved 2021-12-11.
- S2CID 235074954.
- ^ ISBN 978-0-19-856946-6.
- ^ a b c "Vitamin B12 Deficiency: Causes, Symptoms, and Treatment". WebMD. Retrieved 2021-09-21.
- ^ ISBN 978-0-7817-6507-7. Retrieved 10 March 2016.
- ^ a b c Schick P, Conrad ME, Besa EC, eds. (2015-08-15). "Pernicious Anemia, Clinical Presentation". Medscape. Archived from the original on 17 November 2012. Retrieved 21 January 2013.
- ^ a b "Harmful and Sneaky – Vitamin B12 deficiency | Unjury Protein". unjury.com. 15 October 2019. Retrieved 2021-09-21.
- ^ ISBN 978-0-7817-5007-3.
- PMID 10727590.
- PMID 33204422.
- PMID 17043022.
- ^ "Vitamin B12 or folate deficiency anaemia – Complications". nhs.uk. 2017-10-20. Retrieved 2021-10-13.
- ^ Gersten T (2016). "Pernicious anemia". In Zieve D, Ogilvie I, et al. (eds.). MedlinePlus Medical Encyclopedia. Washington, DC: VeriMed Healthcare Network. Archived from the original on 11 March 2016. Retrieved 11 March 2016 – via U.S. National Library of Medicine.
- PMID 31656431.
- PMID 8159105.
- PMID 35109910.
- S2CID 21893726.
- ^ PMID 21296191.
- ^ "Pernicious Anemia | NHLBI, NIH". www.nhlbi.nih.gov. Retrieved 24 February 2022.
- PMID 35005595.
- ^ PMID 34454714.
- ^ PMID 19891010.
- PMID 18405134.
- PMID 19202968.
- S2CID 21448167.
- S2CID 245177279.
- ^ PMID 4180811.
- PMID 14279251.
- ^ S2CID 6994133.
- PMID 7427470.
- ^ PMID 16716475.
- PMID 12643357.
- PMID 22254022.
- PMID 15298442.
- ISBN 978-0-323-03903-1.[page needed]
- S2CID 8493711.
- ^ PMID 16916826.
- ^ PMID 10386505.
- ^ S2CID 73781536.
- ISBN 3-13-508601-1
- PMID 16585128.
- ISBN 978-0-8493-8569-8.
- PMID 36678131.
- ^ a b "What is vitamin B12–deficiency anemia?". National Heart, Lung, and Blood Institute. Retrieved 25 February 2022.
- ^ PMID 18606874.
- S2CID 4639220.
- S2CID 57765412.
- S2CID 47006345.
- S2CID 25093505.
- PMID 15189123.
- better source needed]
- ^ PMID 18463283.
- ^ a b "Vitamin B-12 Associated Neurological Diseases: Background, Pathophysiology, Epidemiology". 22 February 2022. Retrieved 4 March 2022.
- PMID 24552327.
- ^ "History of Vitamin B-12 and Pernicious Anemia". 2021-11-16. Archived from the original on 2021-11-16. Retrieved 2021-11-16.
- ^ "The Nobel Prize in Physiology or Medicine 1934". NobelPrize.org. Retrieved 3 March 2022.
- ^ "The Nobel Prize in Chemistry 1964". NobelPrize.org. Retrieved 2021-11-18.
- ISBN 978-0-9818193-8-9
- S2CID 28038786.
- ^ PMID 34913341.
- PMID 21722960.
- ^ Smith L, Mosley J, Ford M, Courtney J (June 2016). "Cyanocobalamin/Salcaprozate Sodium: A Novel Way to Treat Vitamin B12 Deficiency and Anemia". Journal of Hematology Oncology Pharmacy. 6 (2).
External links
- Pernicious anemia at Curlie
