Heparin
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˈhɛpərɪn/ HEP-ər-in |
| AHFS/Drugs.com | Monograph |
| License data | |
| Pregnancy category |
|
subcutaneous injection | |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Erratic |
| Metabolism | Liver |
| Elimination half-life | 1.5 hours |
| Excretion | Urine[2] |
| Identifiers | |
ECHA InfoCard | 100.029.698 |
| Chemical and physical data | |
| Formula | C12H19NO20S3 |
| Molar mass | 593.45 g·mol−1 |
| |
| | |
Heparin, also known as unfractionated heparin (UFH), is a medication and naturally occurring
Common side effects include bleeding, pain at the injection site, and
Heparin is contraindicated for suspected cases of
Heparin appears to be relatively safe for use during
The discovery of heparin was announced in 1916.
History
Heparin was discovered by Jay McLean and William Henry Howell in 1916, although it did not enter clinical trials until 1935.[15] It was originally isolated from dog liver cells, hence its name (ἧπαρ hēpar is Greek for 'liver'; hepar + -in).
McLean was a second-year medical student at Johns Hopkins University, and was working under the guidance of Howell investigating pro-coagulant preparations, when he isolated a fat-soluble phosphatide anticoagulant in canine liver tissue.[16] In 1918, Howell coined the term 'heparin' for this type of fat-soluble anticoagulant. In the early 1920s, Howell isolated a water-soluble polysaccharide anticoagulant, which he also termed 'heparin', although it was different from the previously discovered phosphatide preparations.[17][18] McLean's work as a surgeon probably changed the focus of the Howell group to look for anticoagulants, which eventually led to the polysaccharide discovery.
It had at first been accepted that it was Howell who discovered heparin. However in the 1940s, Jay McLean became unhappy that he had not received appropriate recognition for what he saw as his own discovery. Though relatively discreet about his claim and not wanting to upset his former chief, he gave lectures and wrote letters claiming that the discovery was his. This gradually became accepted as fact, and indeed after his death in 1959, his obituary credited him as being the true discoverer of heparin. This was elegantly restated in 1963 in a plaque unveiled in Johns Hopkins to commemorate the major contribution (of McLean) to the discovery of heparin in 1916 in collaboration with Professor William Henry Howell.[19]
In the 1930s, several researchers were investigating heparin.
Heparin production experienced a break in the 1990s. Until then, heparin was mainly obtained from cattle tissue, which was a by-product of the
Medical use

Heparin acts as an anticoagulant, preventing the formation of clots and extension of existing clots within the blood. While heparin itself does not break down clots that have already formed (unlike
- NSTEMI
- Atrial fibrillation
- Deep-vein thrombosis and pulmonary embolism(both prevention and treatment)
- Other thrombotic states and conditions
- heart surgery
- ECMO circuit for extracorporeal life support
- Hemofiltration
- Indwelling central or peripheral venous catheters
Heparin and its low-molecular-weight derivatives (e.g.,
In angiography, 2 to 5 units/mL of unfractionated heparin saline flush is used as a locking solution to prevent the clotting of blood in guidewires, sheaths, and catheters, thus preventing thrombus from dislodging from these devices into the circulatory system .[27][28]
Unfractionated heparin is used in hemodialysis. Comparing to low-molecular-weight heparin, unfractionated heparin does not have prolonged anticoagulation action after dialysis, and low cost. However, the short duration of action for heparin would require it to maintain continuous infusion to maintain its action. Meanwhile, unfractionated heparin has higher risk of heparin-induced thrombocytopenia.[29]
Adverse effects
A serious side-effect of heparin is heparin-induced thrombocytopenia (HIT), caused by an immunological reaction that makes platelets a target of immunological response, resulting in the degradation of platelets, which causes thrombocytopenia.[30] This condition is usually reversed on discontinuation, and in general can be avoided with the use of synthetic heparins. Not all patients with heparin antibodies will develop thrombocytopenia. Also, a benign form of thrombocytopenia is associated with early heparin use, which resolves without stopping heparin. Approximately one-third of patients with diagnosed heparin-induced thrombocytopenia will ultimately develop thrombotic complications.[31]
Two non-hemorrhagic side-effects of heparin treatment are known. The first is elevation of serum
As with many drugs, overdoses of heparin can be fatal. In September 2006, heparin received worldwide publicity when three prematurely born infants died after they were mistakenly given overdoses of heparin at an Indianapolis hospital.[32]
Contraindications
Heparin is contraindicated in those with risk of bleeding (especially in people with uncontrolled blood pressure, liver disease, and stroke), severe liver disease, or severe hypertension.[33]
Antidote to heparin
Protamine sulfate has been given to counteract the anticoagulant effect of heparin (1 mg per 100 units of heparin that had been given over the past 6 hours).[34] It may be used in those who overdose on heparin or to reverse heparin's effect when it is no longer needed.[35]
Physiological function
Heparin's normal role in the body is unclear. Heparin is usually stored within the secretory granules of
Evolutionary conservation
In addition to the bovine and porcine tissue from which pharmaceutical-grade heparin is commonly extracted, it has also been extracted and characterized from:
The biological activity of heparin within species 6–11 is unclear and further supports the idea that the main physiological role of heparin is not anticoagulation. These species do not possess any blood coagulation system similar to that present within the species listed 1–5. The above list also demonstrates how heparin has been highly evolutionarily conserved, with molecules of a similar structure being produced by a broad range of organisms belonging to many different phyla.[citation needed]
Pharmacology
In
Mechanism of action
Heparin binds to the enzyme inhibitor
- GlcNAc/NS(6S)-GlcA-GlcNS(3S,6S)-IdoA(2S)-GlcNS(6S)
The conformational change in AT on heparin-binding mediates its inhibition of factor Xa. For thrombin inhibition, however, thrombin must also bind to the heparin polymer at a site proximal to the pentasaccharide. The highly negative charge density of heparin contributes to its very strong
This size difference has led to the development of low-molecular-weight heparins (LMWHs) and fondaparinux as anticoagulants. Fondaparinux targets anti-factor Xa activity rather than inhibiting thrombin activity, with the aim of facilitating a more subtle regulation of coagulation and an improved therapeutic index. It is a synthetic pentasaccharide, whose chemical structure is almost identical to the AT binding pentasaccharide sequence that can be found within polymeric heparin and heparan sulfate.
With LMWH and fondaparinux, the risk of
Danaparoid, a mixture of heparan sulfate, dermatan sulfate, and chondroitin sulfate can be used as an anticoagulant in patients having developed HIT. Because danaparoid does not contain heparin or heparin fragments, cross-reactivity of danaparoid with heparin-induced antibodies is reported as less than 10%.[55]
The effects of heparin are measured in the lab by the partial thromboplastin time (
Administration

Heparin is given
The American College of Chest Physicians publishes clinical guidelines on heparin dosing.[58]
Natural degradation or clearance
Unfractionated heparin has a half-life of about one to two hours after infusion,[56] whereas low-molecular-weight heparin's half-life is about four times longer. Lower doses of heparin have a much shorter half-life than larger ones. Heparin binding to macrophage cells is internalized and depolymerized by the macrophages. It also rapidly binds to endothelial cells, which precludes the binding to antithrombin that results in anticoagulant action. For higher doses of heparin, endothelial cell binding will be saturated, such that clearance of heparin from the bloodstream by the kidneys will be a slower process.[59]
Chemistry
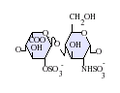
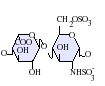
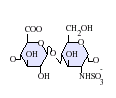
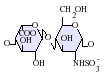
Heparin structure

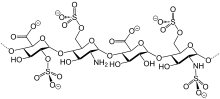
Native heparin is a polymer with a
Not shown below are the rare disaccharides containing a 3-O-sulfated glucosamine (GlcNS(3S,6S)) or a free amine group (GlcNH3+). Under physiological conditions, the ester and amide sulfate groups are deprotonated and attract positively charged counterions to form a heparin salt. Heparin is usually administered in this form as an anticoagulant.
-
IdoA(2S)-GlcNS(6S)*
-
IdoA(2S)-GlcNS
-
IdoA-GlcNS(6S)
-
GlcA-GlcNAc
-
GlcA-GlcNS
-
IdoA-GlcNS
GlcA = β-D-glucuronic acid, IdoA = α-L-iduronic acid, IdoA(2S) = 2-O-sulfo-α-L-iduronic acid, GlcNAc = 2-deoxy-2-acetamido-α-D-glucopyranosyl, GlcNS = 2-deoxy-2-sulfamido-α-D-glucopyranosyl, GlcNS(6S) = 2-deoxy-2-sulfamido-α-D-glucopyranosyl-6-O-sulfate
One unit of heparin (the "Howell unit") is an amount approximately equivalent to 0.002 mg of pure heparin, which is the quantity required to keep 1 ml of cat's blood fluid for 24 hours at 0 °C.[63]
Three-dimensional structure
The three-dimensional structure of heparin is complicated because iduronic acid may be present in either of two low-energy conformations when internally positioned within an oligosaccharide. The conformational equilibrium is influenced by sulfation state of adjacent glucosamine sugars.[64] Nevertheless, the solution structure of a heparin dodecasaccharide composed solely of six GlcNS(6S)-IdoA(2S) repeat units has been determined using a combination of NMR spectroscopy and molecular modeling techniques.[65] Two models were constructed, one in which all IdoA(2S) were in the 2S0 conformation (A and B below), and one in which they are in the 1C4 conformation (C and D below). However, no evidence suggests that changes between these conformations occur in a concerted fashion. These models correspond to the protein data bank code 1HPN.[66]
In the image above:
- A = 1HPN (all IdoA(2S) residues in 2S0 conformation) Jmol viewer
- B = van der Waals radius space filling model of A
- C = 1HPN (all IdoA(2S) residues in 1C4 conformation) Jmol viewer
- D = van der Waals radius space filling model of C
In these models, heparin adopts a helical conformation, the rotation of which places clusters of sulfate groups at regular intervals of about 17
Depolymerization techniques
Either chemical or enzymatic depolymerization techniques or a combination of the two underlie the vast majority of analyses carried out on the structure and function of heparin and heparan sulfate (HS).
Enzymatic
The enzymes traditionally used to digest heparin or HS are naturally produced by the soil bacterium Pedobacter heparinus (formerly named Flavobacterium heparinum).[67] This bacterium is capable of using either heparin or HS as its sole carbon and nitrogen source. To do so, it produces a range of enzymes such as lyases, glucuronidases, sulfoesterases, and sulfamidases.[68] The lyases have mainly been used in heparin/HS studies. The bacterium produces three lyases, heparinases I (EC 4.2.2.7), II (no EC number assigned) and III (EC 4.2.2.8) and each has distinct substrate specificities as detailed below.[69][70]
| Heparinase enzyme | Substrate specificity |
Heparinase I |
GlcNS(±6S)-IdoA(2S) |
| Heparinase II | GlcNS/Ac(±6S)-IdoA(±2S) GlcNS/Ac(±6S)-GlcA |
Heparinase III |
GlcNS/Ac(±6S)-GlcA/IdoA (with a preference for GlcA) |

The lyases cleave heparin/HS by a
Chemical
Nitrous acid can be used to chemically depolymerize heparin/HS. Nitrous acid can be used at pH 1.5 or at a higher pH of 4. Under both conditions, nitrous acid effects deaminative cleavage of the chain.[73]

At both 'high' (4) and 'low' (1.5) pH, deaminative cleavage occurs between GlcNS-GlcA and GlcNS-IdoA, albeit at a slower rate at the higher pH. The deamination reaction, and therefore chain cleavage, is regardless of O-sulfation carried by either monosaccharide unit.
At low pH, deaminative cleavage results in the release of inorganic SO4, and the conversion of GlcNS into anhydromannose (aMan). Low-pH nitrous acid treatment is an excellent method to distinguish N-sulfated polysaccharides such as heparin and HS from non N-sulfated polysaccharides such as chondroitin sulfate and dermatan sulfate, chondroitin sulfate and dermatan sulfate not being susceptible to nitrous acid cleavage.
Detection in body fluids
Current clinical laboratory assays for heparin rely on an indirect measurement of the effect of the drug, rather than on a direct measure of its chemical presence. These include
Other functions
- Blood specimen test tubes, EDTA of not affecting levels of most ions. However, the concentration of ionized calcium may be decreased if the concentration of heparin in the blood specimen is too high.[76] Heparin can interfere with some immunoassays, however. As lithium heparin is usually used, a person's lithium levels cannot be obtained from these tubes; for this purpose, royal-blue-topped (and dark green-topped) vacutainers containing sodiumheparin are used.
- Heparin-coated blood oxygenators are available for use in heart-lung machines. Among other things, these specialized oxygenators are thought to improve overall biocompatibilityand host homeostasis by providing characteristics similar to those of native endothelium.
- The DNA binding sites on RNA polymerase can be occupied by heparin, preventing the polymerase from binding to promoter DNA. This property is exploited in a range of molecular biological assays.
- Common diagnostic procedures require PCR amplification of a patient's DNA, which is easily extracted from white blood cells treated with heparin. This poses a potential problem, since heparin may be extracted along with the DNA, and it has been found to interfere with the PCR reaction at levels as low as 0.002 U in a 50 μL reaction mixture.[77]
- Heparin has been used as a pseudotype retroviral and lentiviral vectors for gene therapy, allows it to be used for downstream purification of viral vectors.[83][84]
- Heparin is being trialed in a nasal spray form as prophylaxis against COVID-19 infection.[85] Furthermore, its reported from trials that due to anti-viral, anti-inflammatory and its anti-clotting effects its inhalation could improve at a 70% rate on patients that were actively struck by a COVID-19 infection.[86]
Society and culture
Contamination recalls
Considering the animal source of pharmaceutical heparin, the numbers of potential impurities are relatively large compared with a wholly synthetic therapeutic agent. The range of possible biological contaminants includes viruses, bacterial endotoxins, transmissible spongiform encephalopathy (TSE) agents, lipids, proteins, and DNA. During the preparation of pharmaceutical-grade heparin from animal tissues, impurities such as solvents, heavy metals, and extraneous cations can be introduced. However, the methods employed to minimize the occurrence and to identify and/or eliminate these contaminants are well established and listed in guidelines and pharmacopoeias. The major challenge in the analysis of heparin impurities is the detection and identification of structurally related impurities. The most prevalent impurity in heparin is dermatan sulfate (DS), also known as chondroitin sulfate B. The building-block of DS is a disaccharide composed of 1,3-linked N-acetyl galactosamine (GalN) and a uronic acid residue, connected via 1,4 linkages to form the polymer. DS is composed of three possible uronic acid (GlcA, IdoA or IdoA2S) and four possible hexosamine (GalNAc, Gal- NAc4S, GalNAc6S, or GalNAc4S6S) building-blocks. The presence of iduronic acid in DS distinguishes it from chrondroitin sulfate A and C and likens it to heparin and HS. DS has a lower negative charge density overall compared to heparin. A common natural contaminant, DS is present at levels of 1–7% in heparin API, but has no proven biological activity that influences the anticoagulation effect of heparin.[87]
In December 2007, the
2008 recall due to adulteration in drug from China
In March 2008, major recalls of heparin were announced by the FDA due to contamination of the raw heparin stock imported from China.[89][90] According to the FDA, the adulterated heparin killed nearly 80 people in the United States.[91] The adulterant was identified as an "over-sulphated" derivative of chondroitin sulfate, a popular shellfish-derived supplement often used for arthritis, which was intended to substitute for actual heparin in potency tests.[92]
According to the New York Times: "Problems with heparin reported to the agency include difficulty breathing, nausea, vomiting, excessive sweating and rapidly falling blood pressure that in some cases led to life-threatening shock".
Use in homicide
In 2006, Petr Zelenka, a nurse in the Czech Republic, deliberately administered large doses to patients, killing seven, and attempting to kill ten others.[93]
Overdose issues
In 2007, a nurse at
In July 2008, another set of twins born at Christus Spohn Hospital South, in Corpus Christi, Texas, died after an accidentally administered overdose of the drug. The overdose was due to a mixing error at the hospital pharmacy and was unrelated to the product's packaging or labeling.[99] As of July 2008[update], the exact cause of the twins' death was under investigation.[100][101]
In March 2010, a two-year-old transplant patient from Texas was given a lethal dose of heparin at the University of Nebraska Medical Center. The exact circumstances surrounding her death are still under investigation.[102]
Production
Pharmaceutical-grade heparin is derived from
Research
As detailed in the table below, the potential is great for the development of heparin-like structures as drugs to treat a wide range of diseases, in addition to their current use as anticoagulants.[106][107]
| Disease states sensitive to heparin | Heparin's effect in experimental models | Clinical status |
Acquired immunodeficiency syndrome |
Reduces the ability of human immunodeficiency virus types 1 and 2 to adsorb to cultured T4 cells.[108] |
– |
Adult respiratory distress syndrome |
Reduces cell activation and accumulation in airways, neutralizes mediators and cytotoxic cell products, and improves lung function in animal models | Controlled clinical trials |
| Allergic encephalomyelitis | Effective in animal models |
– |
| Allergic rhinitis | Effects as for adult respiratory distress syndrome, although no specific nasal model has been tested | Controlled clinical trial |
| Arthritis | Inhibits cell accumulation, collagen destruction and angiogenesis | Anecdotal report
|
| Asthma | As for adult respiratory distress syndrome, however, it has also been shown to improve lung function in experimental models | Controlled clinical trials |
| Cancer | Inhibits tumour growth, metastasis and angiogenesis, and increases survival time in animal models |
Several anecdotal reports |
| Delayed-type hypersensitivity reactions | Effective in animal models | – |
| Inflammatory bowel disease | Inhibits inflammatory cell transport in general, no specific model tested | Controlled clinical trials |
| Interstitial cystitis | Effective in a human experimental model of interstitial cystitis | Related molecule now used clinically |
| Transplant rejection | Prolongs allograft survival in animal models |
– |
- – indicates that no information is available
As a result of heparin's effect on such a wide variety of disease states, a number of drugs are indeed in development whose molecular structures are identical or similar to those found within parts of the polymeric heparin chain.[106]
| Drug molecule | Effect of new drug compared to heparin | Biological activities |
| Heparin tetrasaccharide | Nonanticoagulant, nonimmunogenic, orally active | Antiallergic |
| Pentosan polysulfate | Plant derived, little anticoagulant activity, anti-inflammatory, orally active | Anti-inflammatory, antiadhesive, antimetastatic |
| Phosphomannopentanose sulfate | Potent inhibitor of heparanase activity | Antimetastatic, antiangiogenic, anti-inflammatory |
| Selectively chemically O-desulphated heparin | Lacks anticoagulant activity | Anti-inflammatory, antiallergic, antiadhesive |
References
- ^ a b "Heparin Interpharma APMDS". Therapeutic Goods Administration (TGA). 7 December 2023. Retrieved 7 March 2024.
- ^ "Heparin". 10 February 2012. Archived from the original on 14 February 2012.
- ^ a b c d e f "Heparin Sodium". The American Society of Health-System Pharmacists. Archived from the original on 27 January 2016. Retrieved 1 January 2016.
- ^ a b "Heparin (Mucous) Injection BP – Summary of Product Characteristics". Electronic Medicines Compendium. September 2016. Archived from the original on 20 December 2016. Retrieved 15 December 2016.
- PMID 23687625.
- ISBN 978-0-683-30751-1. Archivedfrom the original on 10 September 2017.
- ^ "AstraZeneca COVID-19-Vakzine Umgang mit dem Risiko von Gerinnungskomplikationen" (PDF). Archived (PDF) from the original on 13 January 2024. Retrieved 3 April 2021.
- PMID 33835769.
- S2CID 233663558. Archived (PDF) from the original on 30 March 2021. Retrieved 3 April 2021.)
{{cite journal}}: Cite journal requires|journal=(help - ^ "Heparin Pregnancy and Breastfeeding Warnings". drugs.com. Archived from the original on 27 January 2016. Retrieved 15 January 2016.
- ISBN 978-0-7216-0240-0.
- ISBN 978-1-118-35446-9. Archivedfrom the original on 10 September 2017.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ISBN 978-1-55009-378-0. Archivedfrom the original on 10 September 2017.
- ^ "Heparin used as an anticoagulant". AnimalResearch.info. Archived from the original on 23 October 2013.
- PMID 13619023.
- ^ Howell WH (1922). "Heparin, an anticoagulant". American Journal of Physiology. 63: 434–435.
- PMID 8281678.
- ^ "The discovery of heparin - Hektoen International". hekint.org. 21 March 2024. Retrieved 30 March 2024.
- PMID 16745848.
- ^ Rutty CJ. "Miracle Blood Lubricant: Connaught and the Story of Heparin, 1928–1937". Health Heritage Research Services. Archived from the original on 23 August 2007. Retrieved 21 May 2007.
- S2CID 246575794.
- ^ PMID 30855835. Retrieved 31 October 2022.
- PMID 9654538.
- PMID 11919306.
- PMID 12519540.
- S2CID 225587226.
- PMID 27190678.
- PMID 21039876.
- PMID 17823223.
- S2CID 22963756.
- ^ Kusmer K (20 September 2006). "3rd Ind. preemie infant dies of overdose". Fox News. Associated Press. Archived from the original on 18 October 2007. Retrieved 8 January 2007.
- ^ Australian Medicines Handbook (2019 (online) ed.). Adelaide: Australian Medicines Handbook Pty Ltd. January 2019.
- ^ Internal medicine, Jay H. Stein, p. 635
- ^ "Protamine Sulfate". The American Society of Health-System Pharmacists. Archived from the original on 6 November 2016. Retrieved 8 December 2016.
- PMID 10412563.
- ^ ISBN 978-0-7167-4339-2.
- PMID 12524047.
- PMID 7287679.
- PMID 15012907.
- PMID 6807978.
- PMID 1463730.
- PMID 6213614.
- S2CID 42108805. Archived from the originalon 27 September 2007. Retrieved 22 March 2007.
- PMID 3624220.
- PMID 10434045.
- ^ PMID 10913828.
- PMID 20937523.
- S2CID 671954.
- PMID 18777207.
- PMID 16030035.
- PMID 11278930.
- S2CID 29785682.
- S2CID 4339441.
- ^ Shalansky, Karen. DANAPAROID (Orgaran) for Heparin-Induced Thrombocytopenia. Archived 28 September 2007 at the Wayback Machine Vancouver Hospital & Health Sciences Centre, February 1998 Drug & Therapeutics Newsletter. Retrieved on 8 January 2007.
- ^ from the original on 9 September 2011.
- PMID 15339877.
- PMID 15383472.
- S2CID 33367673.
- ISBN 978-0-07-143591-8. Archived from the originalon 7 July 2011.
- .
- ISSN 0024-9297.
- ^ "Online Medical Dictionary". Centre for Cancer Education. 2000. Archived from the original on 13 August 2007. Retrieved 11 July 2008.
- PMID 2331699.
- PMID 8352752.
- ^ Mulloy B, Forster MJ. "N.M.R. and molecular-modeling studies of the solution conformation of heparin". Archived from the original on 11 October 2008. Retrieved 17 July 2006.
- PMID 16565082.
- PMID 7235692.
- PMID 2334685.
- PMID 8347612.
- PMID 5062409.
- PMID 3196292.
- PMID 9127.
- S2CID 44678237.
- ISBN 978-0-9626523-7-0.
- PMID 18018679. Archived from the original(PDF) on 10 September 2016. Retrieved 18 April 2016.
- PMID 10323479.
- PMID 18369865.
- ^ "Affinity Chromatography". Sigma-Aldrich. Archived from the original on 7 May 2016.
- ^ "HiTrap Heparin HP". GE Healthcare Life Sciences. Archived from the original on 1 August 2017.
- ^ "Performing a Separation of DNA binding proteins with GE Healthcare Products Based on Heparin". Sigma-Aldrich. Archived from the original on 16 April 2019. Retrieved 16 April 2019.
- PMID 11991744.
- PMID 18470635.
- PMID 15812800.
- ^ Rollason B (21 December 2021). "Melbourne researchers trial use of common blood-thinning drug heparin to combat COVID-19". ABC News. Archived from the original on 22 December 2021. Retrieved 22 December 2021.
- ^ Margo J (19 January 2022). "A 70-year-old drug may be the answer to treating COVID-19". The Australian Financial Review. Archived from the original on 22 January 2022. Retrieved 22 January 2022.
- PMID 20814668.
- ^ "AM2 PAT, Inc. Issues Nationwide Recall of Pre-Filled Heparin Lock Flush Solution USP (5 mL in 12 mL Syringes)" (Press release). US FDA. 20 December 2007. Archived from the original on 23 December 2007.
- ^ CBS News. "Blood-thinning drug under suspicion". Archived from the original on 23 October 2012.
- ^ "Information on Heparin" (Press release). US FDA. Archived from the original on 15 April 2012.
- ^ Darby N (18 September 2018). "The Past And Future Of Managing Raw Material And Process Risks In Biomanufacturing". Drug Discovery Online. VertMarkets. A Supply Chain Under Scrutiny. Archived from the original on 1 November 2018. Retrieved 1 November 2018.
- U.S. Food and Drug Administration. Archived(PDF) from the original on 6 March 2010. Retrieved 23 April 2008.
- ^ Velinger J (12 May 2006). "Nurse committed murders to "test" doctors". Radio Praha. Archived from the original on 24 September 2009.
- ^ Ornstein C, Gorman A (21 November 2007). "Report: Dennis Quaid's twins get accidental overdose". Los Angeles Times. Archived from the original on 7 March 2008.
- ^ Jablon R (4 December 2007). "Dennis Quaid and wife sue drug maker". USA Today. Archived from the original on 28 June 2010.
- ^ Ornstein C (5 December 2007). "Dennis Quaid files suit over drug mishap". Los Angeles Times. Archived from the original on 4 July 2008.
- ^ "Quaid Awarded $750,000 Over Hospital Negligence". SFGate.com. 16 December 2008. Archived from the original on 15 April 2009.
- ^ Sanz A. "Coroner's office investigates infant deaths". WTHR NBC News. Archived from the original on 29 June 2011.
- ^ Statement by Dr. Richard Davis, Chief Medical Officer, CHRISTUS Spohn Health System[permanent dead link], 10 July 2008
- ^ "At a Glance Heparin Overdose at Hospital". Dallas Morning News. 11 July 2008. Archived from the original on 25 October 2008.
- ^ Vonfremd M, Ibanga I (11 July 2008). "Officials Investigate Infants' Heparin OD at Texas Hospital". ABC News. Archived from the original on 11 July 2008. Retrieved 24 July 2008.
- ^ "Nebraska Med. Center Investigates Staff After Girl's Death". KETV Omaha. 31 March 2010. Archived from the original on 20 March 2012.
- PMID 10549711.
- ^ Bhattacharya A (August 2008). "Flask synthesis promises untainted heparin". Chemistry World. Royal Society of Chemistry. Archived from the original on 21 October 2012. Retrieved 6 February 2011.
- PMID 22034431.
- ^ S2CID 7334825.
- S2CID 6380429.
- PMID 2457906.
Further reading
- Marcum JA (January 2000). "The origin of the dispute over the discovery of heparin". Journal of the History of Medicine and Allied Sciences. 55 (1): 37–66. S2CID 30050513.
- Mulloy B, Hogwood J, Gray E, Lever R, Page CP (January 2016). "Pharmacology of Heparin and Related Drugs". Pharmacological Reviews. 68 (1): 76–141. PMID 26672027.