Wave–particle duality
| Part of a series of articles about |
| Quantum mechanics |
|---|
Wave–particle duality is the concept in quantum mechanics that quantum entities exhibit particle or wave properties according to the experimental circumstances.[1]: 59 It expresses the inability of the classical concepts such as particle or wave to fully describe the behavior of quantum objects.[2]: III:1-1 During the 19th and early 20th centuries, light was found to behave as a wave, and then later discovered to have a particulate character, whereas electrons were found to act as particles, and then later discovered to have wavelike aspects. The concept of duality arose to name these contradictions.
History
Wave-particle duality of light
In the late 17th century Sir
Wave-particle duality of matter
The contradictory evidence from electrons arrived in the opposite order. Many experiments by
Following de Broglie's proposal of wave–particle duality of electrons, in 1925 to 1926, Erwin Schrödinger developed the wave equation of motion for electrons. This rapidly became part of what was called by Schrödinger undulatory mechanics,[9] now called the Schrödinger equation and also "wave mechanics".
In 1926, Max Born gave a talk in an Oxford meeting about using the electron diffraction experiments to confirm the wave-particle duality of electrons. In his talk, Born cited experimental data from Clinton Davisson in 1923. It happened that Davisson also attended that talk. Davisson returned to his lab in the US to switch his experimental focus to test the wave property of electrons.[10]
In 1927, the wave nature of electrons was empirically confirmed by two experiments. The Davisson–Germer experiment at Bell Labs measured electrons scattered from Ni metal surfaces.[11][12][13][14][15] George Paget Thomson and Alexander Reid at Cambridge University scattered electrons through thin metal films and observed concentric diffraction rings.[16] Alexander Reid, who was Thomson's graduate student, performed the first experiments,[17] but he died soon after in a motorcycle accident[18] and is rarely mentioned. These experiments were rapidly followed by the first non-relativistic diffraction model for electrons by Hans Bethe[19] based upon the Schrödinger equation, which is very close to how electron diffraction is now described. Significantly, Davisson and Germer noticed[14][15] that their results could not be interpreted using a Bragg's law approach as the positions were systematically different; the approach of Bethe,[19] which includes the refraction due to the average potential, yielded more accurate results. Davisson and Thomson were awarded the Nobel Prize in 1937 for experimental verification of wave property of electrons by diffraction experiments.[20] Similar crystal diffraction experiments were carried out by Otto Stern in the 1930s using beams of helium atoms and hydrogen molecules. These experiments further verified that wave behavior is not limited to electrons and is a general property of matter on a microscopic scale.
Classical waves and particles
Before proceeding further, it is critical to introduce some definitions of waves and particles both in a classical sense and in quantum mechanics. Waves and particles are two very different models for physical systems, each with an exceptionally large range of application. Classical waves obey the
Classical particles obey
Some experiments on quantum systems show wave-like interference and diffraction; some experiments show particle-like collisions.
Quantum systems obey wave equations that predict particle probability distributions. These particles are associated with discrete values called quanta for properties such as spin, electric charge and magnetic moment. These particles arrive one at time, randomly, but build up a pattern. The probability that experiments will measure particles at a point in space is the square of a complex-number valued wave. Experiments can be designed to exhibit diffraction and interference of the probability amplitude.[1] Thus statistically large numbers of the random particle appearances can display wave-like properties. Similar equations govern collective excitations called quasiparticles.
Electrons behaving as waves and particles
The electron double slit experiment is a textbook demonstration of wave-particle duality.[2] A modern version of the experiment is shown schematically in the figure below.

Electrons from the source hit a wall with two thin slits. A mask behind the slits can expose either one or open to expose both slits. The results for high electron intensity are shown on the right, first for each slit individually, then with both slits open. With either slit open there is a smooth intensity variation due to diffraction. When both slits are open the intensity oscillates, characteristic of wave interference.
Having observed wave behavior, now change the experiment, lowering the intensity of the electron source until only one or two are detected per second, appearing as individual particles, dots in the video. As shown in the movie clip below, the dots on the detector seem at first to be random. After some time a pattern emerges, eventually forming an alternating sequence of light and dark bands.
The experiment shows wave interference revealed a single particle at a time -- quantum mechanical electrons display both wave and particle behavior. Similar results have been shown for atoms and even large molecules.[22]
Observing photons as particles
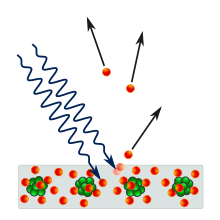
While electrons were thought to be particles until their wave properties were discovered; for photons it was the opposite. In 1887,

where h is the
Both discrete (quantized) energies and also momentum are, classically, particle attributes. There are many other examples where photons display particle-type properties, for instance in solar sails, where sunlight could propel a space vehicle and laser cooling where the momentum is used to slow down (cool) atoms. These are a different aspect of wave-particle duality.
Duality with other properties
Normally angular momentum is considered to be a particle-like property, for instance a spinning top. However, light waves can have angular momentum when they form a vortex,[27] as illustrated in the Figure. Electron waves with angular momentum have also been produced,[28] with up to a thousand angular momentum quanta.[29] These matter waves will diffract and interfere, while simultaneously having particle-like angular momentum so they will interact with magnetic fields, similar to a current loop.
Which slit experiments
In a "which way" experiment, particle detectors are placed at the slits to determine which slit the electron traveled through. When these detectors are inserted, quantum mechanics predicts that the interference pattern disappears because the detected part of the electron wave has changed (loss of coherence).[2] Many similar proposals have been made and many have been converted into experiments and tried out.[30] Every single one shows the same result: as soon as electron trajectories are detected, interference disappears.
A simple example of these "which way" experiments uses a Mach–Zehnder interferometer, a device based on lasers and mirrors sketched below.[31]

A laser beam along the input port splits at a half-silvered mirror. Part of the beam continues straight, passes though a glass
Each output port has a camera to record the results. The two beams show interference characteristic of wave propagation. If the laser intensity is turned sufficiently low, individual dots appear on the cameras, building up the pattern as in the electron example.[31]
The first beam-splitter mirror acts like double slits, but in the interferometer case we can remove the second beam splitter. Then the beam heading down ends up in output port 1: any photon particles on this path gets counted in that port. The beam going across the top ends up on output port 2. In either case the counts will track the photon trajectories. However, as soon as the second beam splitter is removed the interference pattern disappears.[31]
See also
- Basic concepts of quantum mechanics– Non-technical introduction to quantum physics
- Complementarity (physics) – Quantum physics concept
- Einstein's thought experiments
- Interpretations of quantum mechanics
- Wheeler's delayed choice experiment– Number of quantum physics thought experiments
- Uncertainty principle
- Matter wave
- Corpuscular theory of light
References
- ^ ISBN 0486409244.
- ^ ISBN 978-0-201-02118-9.
- ^ Christiaan Huygens, Traité de la lumiere... (Leiden, Netherlands: Pieter van der Aa, 1690), Chapter 1.
- .
- ISBN 978-0-691-10250-4.
- ^ ISBN 978-0-486-26126-3.
- ISSN 1941-5982.
- ^ de Broglie, Louis Victor. "On the Theory of Quanta" (PDF). Foundation of Louis de Broglie (English translation by A.F. Kracklauer, 2004. ed.). Retrieved 25 February 2023.
- ISSN 0031-899X.
- ISSN 0031-9228.
- S2CID 4104602.
- ISSN 0031-899X.
- ISSN 0031-899X.
- ^ PMID 16587341.
- ^ PMID 16587378.
- S2CID 4122313.
- S2CID 98311959.
- S2CID 171025814.
- ^ .
- ^ "The Nobel Prize in Physics 1937". NobelPrize.org. Retrieved 2024-03-18.
- ^ S2CID 832961.
- S2CID 56438353.
- ^ Whittaker, E. T. (1910). A History of the Theories of Aether and Electricity: From the Age of Descartes to the Close of the Nineteenth Century. Longman, Green and Co.
- JSTOR 27757381.
- S2CID 120382028. Archived from the original on September 21, 2020. Retrieved December 14, 2020 – via Random House Audiobooks. Alt URL.
- doi:10.1002/andp.19053220607, translated into English as Einstein, A. "On a Heuristic Point of View about the Creation and Conversion of Light" (PDF). The Old Quantum Theory. Archived from the original(PDF) on 11 June 2009. The term "photon" was introduced in 1926.
- PMID 9906912.
- S2CID 2970408.
- S2CID 247187983.
- S2CID 34901303.
- ^ ISSN 0002-9505.
External links
- R. Nave. "Wave–Particle Duality". HyperPhysics. Georgia State University, Department of Physics and Astronomy. Retrieved December 12, 2005.
- "Wave–particle duality". PhysicsQuest. American Physical Society. Retrieved August 31, 2023.
- Mack, Katie. "Quantum 101 – Quantum Science Explained". Perimeter Institute for Theoretical Physics. Retrieved August 31, 2023.








