White blood cell differential
| White blood cell differential | |
|---|---|
white blood cells in peripheral blood | |
| MedlinePlus | 003657 |
| eMedicine | 2085133 |
| LOINC | 33255-1, 24318-8, 69738-3 |
A white blood cell differential is a
White blood cell differentials may be performed by an
In 1674, Antonie van Leeuwenhoek published the first microscopic observations of blood cells. Improvements in microscope technology throughout the 18th and 19th centuries allowed the three cellular components of blood to be identified and counted. In the 1870s, Paul Ehrlich invented a staining technique that could differentiate between each type of white blood cell. Dmitri Leonidovich Romanowsky later modified Ehrlich's stain to produce a wider range of colours, creating the Romanowsky stain, which is still used to stain blood smears for manual differentials.
Automation of the white blood cell differential began with the invention of the Coulter counter, the first automated hematology analyzer, in the early 1950s. This machine used electrical impedance measurements to count cells and determine their sizes, allowing white and red blood cells to be enumerated. In the 1970s, two techniques were developed for performing automated differential counts: digital image processing of microscope slides and flow cytometry techniques using light scattering and cell staining. These methods remain in use on modern hematology analyzers.
| Cell type | Adult reference range (× 109/L) |
Adult reference range (percentage) |
|---|---|---|
| Neutrophils | 1.7–7.5 | 50–70 |
| Lymphocytes | 1.0–3.2 | 18–42 |
| Monocytes | 0.1–1.3 | 2–11 |
| Eosinophils | 0.0–0.3 | 1–3 |
| Basophils | 0.0–0.2 | 0–2 |
The white blood cell differential is a common blood test that is often ordered alongside a
Five types of white blood cells are normally found in blood:
CBC and differential testing is usually performed on
Types
Manual differential

In a manual differential, a stained
Procedure

A blood smear is prepared by placing a drop of blood on a microscope slide and using a second slide held at an angle to spread the blood and pull it across the slide, forming a "feathered edge" consisting of a single layer of cells at the end of the smear.[19] This may be done by hand or using an automated slide maker coupled to a hematology analyzer. The slide is treated with a Romanowsky stain, commonly Wright's stain or Wright-Giemsa, and examined under the microscope. The smear is examined in a systematic pattern, scanning from side to side within the feathered edge and counting cells consecutively. The differential is typically performed at 400x or 500x magnification, but 1000x magnification may be used if abnormal cells are present.[20] Cells are identified based on their morphologic features, such as the size and structure of their nucleus and the colour and texture of their cytoplasm. This allows abnormal cell types and changes in cellular appearance to be identified.[8] In most cases, the microscopist counts 100 white blood cells, but 200 may be counted for better representation if the white blood cell count is high.[21] The manual differential count produces percentages of each cell type, which can be multiplied by the total white blood cell count from the analyzer to derive the absolute values.[22]
The manual differential can be partially automated with digital microscopy software,
Limitations
Because relatively few cells are counted in the manual differential, the variability is higher than in automated techniques, especially when cells are present in low amounts.[27] For example, in a sample containing 5 percent monocytes, the manual differential results could be between 1 and 10 percent due to sampling variation.[28] Additionally, cell identification is subjective and the accuracy depends on the skills of the person reading the slide.[27][29] Poor blood smear preparation can cause an uneven distribution of white blood cells, resulting in inaccurate counting,[30] and improper staining can impede cell identification.[31] Overall, manual differential counts exhibit coefficients of variation (CVs) ranging from 5 to 10 percent, while automated differential counts of normal neutrophils and lymphocytes have CVs of about 3 percent.[31]
In
Automated differential
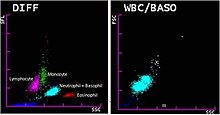
Most
Procedure

Common techniques used by hematology analyzers to identify cells include light scattering,
A small volume of blood (as low as 150 microliters) is aspirated into the analyzer, where reagents are applied to lyse red blood cells and preserve white blood cells. The sample is diluted and passed into a flow cell, which uses hydrodynamic focusing to isolate single cells for accurate analysis of their properties. Various cellular parameters, such as size, complexity and staining reactions, are measured and analyzed to identify cell populations. Basophils are often quantified using a reagent that lyses the cytoplasm of other white blood cells but leaves basophils intact.[31] Samples that have abnormal results[1] or are suspected to contain abnormal cells are flagged by the analyzer for manual blood smear review.[35]
To ensure that results from the automated analyzer are correct, quality control samples are run at least once per day. These are samples with known results that are most often provided by the instrument manufacturer. Laboratories compare their differential results to the known values to ensure the instrument is operating correctly. A moving average measurement may also be used, in which the average results for patient samples are measured at certain intervals. Assuming that the characteristics of the patient population remain roughly the same over time, the average should remain constant. Large shifts in the average value can indicate instrument problems.[39][40]
Limitations
When immature or abnormal white blood cells are present, automated differential results may be incorrect, necessitating a manual blood smear review. Overall, 10 to 25 percent of CBC samples are flagged for manual review by the analyzer.[41] Although most abnormal samples are automatically flagged, some may be missed;[42] conversely, analyzers may generate false positive flags when no abnormal cells are present.[41] Hematology laboratories compensate for these issues by requiring a smear review when differential or CBC results fall outside certain numerical thresholds, regardless of the presence of analyzer flags.[1] The sensitivity and specificity of analyzer flagging can be determined by comparing analyzer flags to manual differential results.[40]
The automated basophil count is notoriously unreliable,[11][31] often underestimating counts in basophilia and producing falsely elevated results in the presence of abnormal cells.[43] The manual differential is therefore considered the reference method for these cells.[11][44]
Analyzers may count nucleated red blood cells,
Cell types and result interpretation

- Neutrophil
- Neutrophils are the most common white blood cells in normal adult blood. When stained with a Romanowsky stain, they exhibit a multi-lobed nucleus and pink cytoplasm that contains small purple granules.[46][47]
The neutrophil count is normally higher in newborns and pregnant women than in other groups.
When stimulated by infection or inflammation, neutrophils may develop abnormal features in their cytoplasm, such as toxic granulation, toxic vacuolation and Döhle bodies. These features, which are caused by the release of cytokines,[56] are collectively known as toxic changes.[13]

- Lymphocyte
- Lymphocytes, which are the second most common type of white blood cell in adults, are typically small cells with a round, dark nucleus and a thin strip of pale blue cytoplasm. Some lymphocytes are larger and contain a few blue granules.[57]
Increased lymphocyte counts (
In response to viral infections (especially infectious mononucleosis), lymphocytes may increase greatly in size, developing unusually shaped nuclei and large amounts of dark blue cytoplasm. Such cells are referred to as reactive or atypical lymphocytes[63] and when present they are either commented on or counted separately from normal lymphocytes in the manual differential.[14]

- Monocyte
- Monocytes are large cells with a curved or folded nucleus and finely granulated, grey-blue cytoplasm that often contains vacuoles. Monocytes are the third most common white blood cell after neutrophils and lymphocytes.[64]
Increased monocyte counts (monocytosis) are seen in chronic infection and inflammation. Extremely high monocyte counts, as well as immature forms of monocytes, occur in chronic myelomonocytic leukemia and acute leukemias of monocytic origin.[65] Monocyte counts may be decreased (monocytopenia) in individuals who are receiving chemotherapy as well as those with aplastic anemia, severe burns, and AIDS.[66]

- Eosinophil
- Eosinophils have large orange granules in their cytoplasm and bi-lobed nuclei. They are found in low amounts in normal blood.[64]
Elevated eosinophil counts (

- Basophil
- Basophils exhibit large, dark purple granules that often cover the cell's nucleus. They are the rarest of the five normal cell types.[69]

- Band neutrophil
A left shift, meaning an increase in band neutrophils or immature granulocytes, can indicate infection, inflammation or bone marrow disorders, although it can also be a normal finding in pregnancy.[71][72] Some laboratories do not separate bands from mature neutrophils in the differential count because the classification is highly subjective and unreliable.[14]

- Immature granulocyte
- Immature granulocytes are immature forms of neutrophils and other granulocytes (eosinophils and basophils). This classification consists of metamyelocytes, myelocytes and promyelocytes, which may be enumerated separately in the manual differential or reported together as immature granulocytes (IG) by automated methods.[73] Immature granulocytes are normally found in the bone marrow, but not in peripheral blood.[74]
When present in significant quantities in the blood, immature granulocytes can indicate infection and inflammation,

- Blast cell
- nucleoli.[10]
When seen on the blood smear, blast cells are an abnormal finding and may be indicative of acute leukemia or other serious blood disorders. Rarely, they may be seen in severe cases of left shift. The presence of Auer rods inside blast cells indicates that they are of myeloid origin, which has important implications for leukemia treatment.[10][77] Other morphologic features can provide information about the lineage of blast cells: for example, myeloblasts tend to be large with distinct nucleoli, while lymphoblasts can be smaller with a denser chromatin pattern. However, these features are not diagnostic, and flow cytometry or special staining is generally used to confirm the lineage.[78]

- Other cells
- Various other abnormal cells may be present in the blood in certain conditions. For example, lymphoma cells may be found on the manual differential in some cases of tumour cells are seen on the peripheral blood smear.[81]
History
Before automated cell counters were introduced, cell counts were performed manually; white and red blood cells, and platelets were counted using microscopes.
By the early years of the 20th century, the white blood cell differential had become a common practice in the United States, but difficulties in interpreting the results cast doubt on the test's utility.[87] In 1906,[88] Charles Langdon Gibson introduced the Gibson chart, which compared the total white blood cell count against the neutrophil count to distinguish between "pyogenic" and "non-pyogenic" conditions and to predict the severity of infections. Around the same time, Josef Arneth proposed a system of classifying neutrophils by their number of nuclear lobes – termed the "lobe index" or Arneth count – and established a set of reference ranges for neutrophil lobularity. Arneth's analysis of neutrophil segmentation was later found to have limited clinical significance, but the association of hypersegmented neutrophils with vitamin B12 and folate deficiency remains accepted.[89][90][91] Viktor Schilling in 1912 proposed a different classification of neutrophils, separating them into "myelozyten, jugendliche, stabkernige and segmentkernige" – that is, myelocytes, "juveniles" (metamyelocytes), band neutrophils (sometimes called "stabs"), and mature, fully segmented neutrophils – and remarked on the clinical significance of the neutrophilic left shift in conjunction with the white blood cell count and the presence of toxic changes. Schilling's monograph, Das Blutbild und seine klinische Verwertung (The Blood Picture and its Clinical Significance), was translated into English in 1926, and his neutrophil classification system quickly found acceptance in American laboratories.[92][93]
The first automated hematology analyzer, the

After basic cell counting had been automated, the white blood cell differential remained a challenge. Research into automating the differential count began in the 1970s and took two main approaches: digital image processing and flow cytometry. Using technology developed in the 1950s and 60s to automate the reading of
Early flow cytometry devices shot beams of light at cells in specific wavelengths and measured the resulting absorbance, fluorescence or light scatter, collecting information about the cells' features and allowing cellular contents such as
See also
References
- ^ PMID 23301216.
- ^ Cornell University College of Veterinary Medicine. "Leukogram". eClinpath. Retrieved 9 August 2019.
- S2CID 110787482.
- ^ Keohane, E et al. (2015). Front matter.
- ^ "Blood Differential: MedlinePlus Lab Test Information". MedlinePlus. Archived from the original on 13 August 2019. Retrieved 12 August 2019.
- ^ American Association for Clinical Chemistry (1 May 2019). "WBC Differential". Lab Tests Online. Archivedfrom the original on 14 April 2019. Retrieved 8 July 2019.
- ^ Kottke-Marchant, K; Davis, B (2012). p. 33.
- ^ a b Turgeon, ML (2016). p. 303.
- ^ PMID 16024331.
- ^ a b c d'Onofrio, G et al. (2014). p. 289.
- ^ PMID 25676369.
- ^ Bain, B et al. (2012). pp. 95–7.
- ^ a b Ciesla, B (2018). p. 153.
- ^ S2CID 4649418.
- ^ Bain, B et al. (2012). pp. 2–4.
- ^ Smock, KJ. Chapter 1 in Greer, JP et al. ed. (2018), sec. "Specimen collection".
- ^ Keohane, E et al. (2015). p. 118.
- ^ Warekois, R; Robinson, R. (2013). p. 116.
- ^ Turgeon, ML. (2016). pp. 346–8.
- ^ Wang, S; Hasserjian, R. (2018). pp. 10–1.
- ^ Wang, S; Hasserjian, R. (2018). p. 10.
- ^ Turgeon, ML. (2016). p. 329.
- ^ Turgeon, ML. (2016). p. 318.
- ^ Bain, B et al. (2012). pp. 44–5.
- ^ PMID 31046197.
- PMID 25676375.
- ^ a b c d Smock, KJ. Chapter 1 in Greer, JP et al. ed. (2018), sec. "Cell counts".
- ^ Ruemke, C. L. (1978). "The statistically expected variability in differential leukocyte counting" (PDF). In Koepke, J. A. (ed.). Differential leukocyte counting. College of American Pathologists Conference / Aspen 1997. Skokie, Illinois: College of American Pathologists.
- ^ McClatchey, K (2002). p. 809.
- ^ Bain, B et al. (2012). pp. 30–1.
- ^ a b c d e Smock, KJ. Chapter 1 in Greer, JP et al. ed. (2018). sec. "Leukocyte differentials".
- ^ Harmening, D (2009). p. 336.
- ^ Wang, S; Hasserjian, R. (2018). p. 9.
- ^ a b Harmening, D (2009). p. 795.
- ^ a b c Bain, B et al. (2012). p. 43.
- ^ Harmening, D (2009). pp. 795–803.
- ^ Naiem, F et al. (2009). p. 210.
- PMID 24797912.
- ^ Kottke-Marchant, K; Davis, B (2012). pp. 697–8.
- ^ PMID 27161194.
- ^ a b Smock, KJ. Chapter 1 in Greer, JP et al ed. (2018), sec. "Advantages and sources of error with automated hematology".
- ^ Bain, B et al. (2012). pp. 43–4.
- PMID 18550479.
- ^ Gibbs, B (2014). p. 88.
- ^ Keohane, E et al. (2015). p. 226.
- ^ Harmening, D (2009). p. 306.
- ^ Bain, B et al. (2012). pp. 88–93.
- ^ Porwit, A et al. (2011). p. 252.
- ^ a b Turgeon, ML. (2016). p. 306.
- ^ Porwit, A et al. (2011). p. 253.
- ^ Hoffman, R et al. (2013). p. 644.
- ^ Porwit, A et al. (2011). pp. 247–52.
- ^ Porwit, A et al. (2011). p. 8.
- PMID 31255364.
- ^ Harmening, D (2009). pp. 308–11
- ^ Hematology and Clinical Microscopy Committee (2019) p. 4.
- ^ Turgeon, ML. (2016). pp. 308–9.
- ^ Turgeon, ML (2016). p. 309.
- ^ a b Porwit, A et al. (2011). p. 258–9.
- PMID 27151266.
- PMID 29261864. Retrieved 24 July 2019.
- ^ Territo, Mary (2018). "Lymphocytopenia". Merck Manuals Professional Edition. Archived from the original on 10 October 2018. Retrieved 22 July 2019.
- ^ Bain, B et al. (2012). pp. 95–7.
- ^ a b Turgeon, ML. (2016). p. 307.
- ^ Bain, B et al. (2012). p. 95.
- ^ Porwit, A et al. (2011). p. 258.
- ^ Bain, B et al. (2012). p. 94.
- ^ a b Porwit, A et al. (2011). p. 256.
- ^ a b Bain, B et al. (2012). pp. 94–5.
- ^ Porwit, A et al. (2011). p. 257.
- ^ a b Bain, B et al. (2012). p. 93.
- ^ Ciesla, B. (2018). pp. 153–4.
- ^ Bain, B et al. (2012). p. 44.
- ^ a b Curry, Choladda Vejabhuti; Staros, Eric (14 January 2015). "Differential Blood Count". EMedicine. Archived from the original on 12 July 2019. Retrieved 17 July 2019.
- ^ Emadi, Ashkan; Law, Jennie (2018). "Chronic Myeloid Leukemia (CML)". Merck Manuals Professional Edition. Archived from the original on 18 August 2019. Retrieved 30 August 2019.
- PMID 26414475.
- ^ Glassy, E. (1998). pp. 6–7.
- ^ Hematology and Clinical Microscopy Committee (2019). pp. 13–22.
- ^ Glassy, E. (1998). p. 228.
- ^ Glassy, E. (1998). p. 328.
- ^ Pereira, I et al. (2011). p. 185.
- ^ Keohane, E et al. (2015). pp. 1–4.
- ^ Kottke-Marchant, K; Davis, B. (2012). p. 1.
- ^ Wintrobe, MM. (1985). p. 10.
- ^ Kottke-Marchant, K; Davis, B. (2012). pp. 3–4.
- S2CID 37401579.
- ^ Koepke, JA (1977). p. 2.
- PMID 17861781.
- ^ Koepke, JA (1977). pp. 2–3.
- ^ Kottke-Marchant, K; Davis, B (2012). pp. 593–4.
- ^ Arneth, J (1904). Die neutrophilen weissen Blutkörperchen bei Infektionskrankheiten (in German). Fischer.
- ^ Koepke, JA (1977). pp. 3–4.
- ^ Schilling, V (1912). Das Blutbild und seine klinische Verwertung, mit Einschluss der Tropenkrankheiten; kurzgefasste technische, theoretische und praktische Anleitung zur mikroskopischen Blutuntersuchung (in German). Fischer.
- ^ Harmening, D (2009). p. 794.
- ^ S2CID 113694419.
- ^ Groner, W (1995). pp. 12–4.
- S2CID 31055044.
- ^ Da Costa, L (2015). p. 5.
- ^ Groner, W (1995). pp. 12–5.
- PMID 2271793.
- PMID 25676375.
- ^ McCann, SR (2016). p. 193.
- ^ Melamed, M (2001). pp. 5–6.
- ^ Shapiro, HM (2003). pp. 84–5.
- ^ a b Melamed, M. (2001). p. 8.
- PMID 22271369.
- PMID 4137312.
- PMID 11933579.
- ^ Koepke, JA (1977). p. 96.
- ^ Kottke-Marchant, K; Davis, B (2012). pp. 8–9.
Bibliography
- Bain, Barbara; Bates, Imelda; Laffan, Mike (2012). Dacie and Lewis practical haematology. Edinburgh: Elsevier Churchill Livingstone. OCLC 780445109.
- Betty Ciesla (27 November 2018). Hematology in Practice. F.A. Davis. ISBN 978-0-8036-6825-6.
- Gibbs, Bernhard (2014). "The absolute basophil count". Basophils and mast cells : methods and protocols. New York, NY: Humana Press. OCLC 889943963.
- Eric F. Glassy (1998). Color Atlas of Hematology: An Illustrated Field Guide Based on Proficiency Testing. College of American Pathologists. ISBN 978-0-930304-66-9.
- John P. Greer; Daniel A. Arber; Bertil E. Glader; Alan F. List; Robert M. Means; George M. Rodgers (19 November 2018). Wintrobe's Clinical Hematology (14th ed.). Wolters Kluwer Health. ISBN 978-1-4963-6713-6.
- Denise Harmening (2009). Clinical Hematology and Fundamentals of Hemostasis (5th ed.). F. A. Davis Company. ISBN 978-0-8036-1732-2.
- Hematology and Clinical Microscopy Committee (2019). "Blood cell identification" (PDF). Hematology and Clinical Microscopy Glossary. College of American Pathologists. Archived (PDF) from the original on 28 June 2019. Retrieved 28 June 2019.
- Ronald Hoffman; Edward J. Benz Jr.; Leslie E. Silberstein; Helen Heslop; John Anastasi; Jeffrey Weitz (1 January 2013). Hematology: Basic Principles and Practice. Elsevier Health Sciences. ISBN 978-1-4377-2928-3.
- Elaine Keohane; Larry Smith; Jeanine Walenga (20 February 2015). Rodak's Hematology: Clinical Principles and Applications. Elsevier Health Sciences. ISBN 978-0-323-23906-6.
- Koepke, JA (1977). Differential leukocyte counting. College of American Pathologists. ISBN 0-930304-12-8.
- Kandice Kottke-Marchant; Bruce Davis (6 June 2012). Laboratory Hematology Practice. John Wiley & Sons. ISBN 978-1-4443-9857-1.
- McCann, SR (3 March 2016). A History of Haematology: From Herodotus to HIV. OUP Oxford. ISBN 978-0-19-102713-0.
- Kenneth D. McClatchey (2002). "Chapter 40: Peripheral blood and bone marrow assessment". Clinical Laboratory Medicine (2nd ed.). Lippincott Williams & Wilkins. ISBN 978-0-683-30751-1.
- Melamed, Myron (2001). "Chapter 1 A brief history of flow cytometry and sorting". Methods in Cell Biology. Vol. 63 part A. Elsevier. pp. 3–17. PMID 11060834.
- Faramarz Naeim; P. Nagesh Rao; Wayne W. Grody (5 March 2009). Hematopathology: Morphology, Immunophenotype, Cytogenetics, and Molecular Approaches. Academic Press. ISBN 978-0-08-091948-5.
- Giuseppe d'Onofrio; Gina Zini (21 October 2014). "Acute Myeloid Leukaemia". Morphology of Blood Disorders (2nd ed.). Wiley. p. 289. ISBN 978-1-118-44258-6.
- Shapiro, Howard (2003). Practical Flow Cytometry (4 ed.). John Wiley & Sons. ISBN 978-0-471-43403-0.
- Irma Pereira; Tracy I. George; Daniel A. Arber (7 December 2011). "Chapter 20: Nonhematopoietic tumors in the blood". Atlas of Peripheral Blood: The Primary Diagnostic Tool. Lippincott Williams & Wilkins. ISBN 978-1-4511-6366-7.
- Anna Porwit; Jeffrey McCullough; Wendy N Erber (27 May 2011). Blood and Bone Marrow Pathology. Elsevier Health Sciences. ISBN 978-0-7020-4535-6.
- Robin S. Warekois; Richard Robinson (27 December 2013). Phlebotomy: Worktext and Procedures Manual. Elsevier Health Sciences. ISBN 978-0-323-29284-9.
- Turgeon, ML (2016). Linné & Ringsrud's Clinical Laboratory Science: Concepts, Procedures, and Clinical Applications (7 ed.). Elsevier Mosby. ISBN 978-0-323-22545-8.
- Sa A. Wang; Robert P. Hasserjian (4 June 2018). Diagnosis of Blood and Bone Marrow Disorders. Springer. ISBN 978-3-319-20279-2.
- Wintrobe, MM (1985). Hematology, the Blossoming of a Science: A Story of Inspiration and Effort. Lea & Febiger. ISBN 978-0-8121-0961-0.
