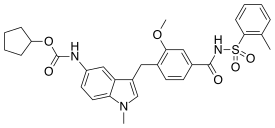
Zafirlukast
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Accolate[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697007 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Unknown |
| Protein binding | >99% (albumin)[1] |
| Metabolism | Hepatic (CYP2C9-mediated) |
| Metabolites | hydroxylated metabolites[1] |
| Elimination half-life | 10 hours |
| Excretion | Fecal[1] |
| Identifiers | |
| |
JSmol) | |
| Melting point | 138 to 140 °C (280 to 284 °F) |
| |
| |
| (verify) | |
Zafirlukast is an orally administered
Zafirlukast, like other LTRAs, works by inhibiting the immune system. Through its action on inflammatory cells in the lungs, zafirlukast reduces the production of inflammatory mediators that are implicated in the pathogenesis of
Zafirlukast (brand name Accolate) was the first cysteinyl leukotriene receptor antagonist approved in the United States. It is now approved in many other countries under other brand names.
Medical uses
Zafirlukast is
Available forms
There are two dosage forms for zafirlukast, notable for their age-adjustments. The 20 mg tablet is for adults and children older than age 12, whereas the 10 mg tablet is for children between the ages of 5 and 12.[1] Tablets should be stored at room temperature, out of direct sunlight, and away from sources of moisture.[1]
Tablets are for oral administration only.[1]
Specific populations
Pediatrics
As a general rule, leukotriene receptor antagonists like zafirlukast are more effective in children that are younger and whose asthma is less
Geriatrics
The hepatic clearance of zafirlukast is impaired in adults 65 years of age and older, resulting in a 2–3 fold increase in the maximum plasma concentration and the total area under the curve. Zafirlukast may increase the risk for infections (7.0% vs 2.9%, zafirlukast vs. placebo incidence respectively), especially lower respiratory tract infections, in older adults, though the infections noted were not severe.[1]
Pregnancy
Zafirlukast is considered to be "pregnancy category B." This is due, in part, to the wide safety margin of zafirlukast in animal studies investigating teratogenicity. No teratogenicity has been observed in doses up to 2000 mg/kg/day in cynomolgus monkeys, representing an equivalent 20x exposure of the maximum recommended daily oral dose in human adults. However, spontaneous abortions occurred in cynomolgus monkeys at 2000 mg/kg/day, though the dose itself was maternally toxic.[1]
Lactation
There is limited research on the use of zafirlukast in women whom are breastfeeding.[5] Based on data from the manufacturer, it is expected that 0.6% of the maternal weight-adjusted dose would reach a breastfed infant, though the effects in the infant are unknown.[5]
Renal impairment
Renal impairment does not appear to affect the pharmacokinetic profile of zafirlukast.[1]
Hepatic impairment
The hepatic clearance of zafirlukast is impaired by significant hepatic impairment. Cirrhosis of the liver can result in an increase in the maximum plasma concentration and the total area under the curve (a measure of drug exposure) by 50–60%.[1]
Contraindications
Zafirlukast is contraindicated in people that are hypersensitive or allergic to it.[1]
Adverse effects
Zafirlukast is generally well tolerated, though headache and gastrointestinal (GI) upset can occur. The incidence of headache is between 12 and 20%, which is similar to the incidence of headache found in patients taking
Other common side effects include flu-like symptoms, sleep disturbances (abnormal dreams, insomnia), hallucinations, and daytime drowsiness.[6]
Neuropsychiatric effects
Neuropsychiatric side effects have been reported with the use of zafirlukast and other LTRAs. While some side effects are less severe (e.g. abnormal dreams), others are more serious (e.g. hallucinations, tremor, suicidality). These effects were discovered through post-marketing reports, as the initial trials were not designed to monitor for neuropsychiatric side effects.[7]
Hepatotoxicity
Zafirlukast can also cause rare but serious side effects like acute liver injury.
According to the "Dear Health Care Provider" letter from AstraZeneca, zafirlukast-induced hepatotoxicity has occurred predominantly in females.[9]
Churg-Strauss syndrome
Several cases of
Overdose
The highest overdose reported with zafirlukast is 200 mg. All overdose patients have survived. Symptoms reported included rash and upset stomach.[1]
Interactions
Drug–drug interactions
Zafirlukast is an inhibitor of the hepatic drug-metabolizing enzyme cytochrome P450 family 3 subfamily A member 4 (CYP3A4).[1] Zafirlukast may increase the concentration of drugs that are metabolized through CYP3A4, such as the anticoagulant medication warfarin and the antiepileptic drugs phenytoin and carbamazepine.[1]
Drug-food interactions
The oral absorption (bioavailability) of zafirlukast is decreased by 40% when it is taken with high fat or high protein meals.[1] To avoid this interaction, zafirlukast should be taken on an empty stomach.[6] An empty stomach is classified as an hour before, or two hours after, consuming a meal.[1]
Pharmacology
Pharmacodynamics
Zafirlukast is an antagonist of
Pharmacokinetics
Absorption
Zafirlukast is rapidly absorbed into the bloodstream following oral administration, reaching peak plasma levels within 3 hours of taking the dose.[1] The peak plasma level is the maximum concentration of zafirlukast in the blood.[14]
Distribution
Zafirlukast is moderately distributed into the body's tissues, with an apparent
Metabolism
Zafirlukast undergoes extensive hepatic metabolism into inactive metabolites.[1] Zafirlukast is primarily metabolized by the enzyme CYP2C9 to a hydroxylated metabolite.[1]
Elimination
Zafirlukast is primarily cleared through biliary excretion at a rate of 20 liters/hour. Zafirlukast is undetectable in urine. The mean terminal half-life ranges 8–16 hours, following linear kinetics up to doses of 80 mg.[1]
Pharmacogenomics
LTC4 synthase
Genetic polymorphisms in the
CYP2C9
Zafirlukast is metabolized through the hepatic enzyme
Chemistry
Synthesis
Zafirlukast can be synthesized by the following method:[20]
Physiochemical properties
Pure zafirlukast is described as a fine, white to pale yellow, amorphous powder. It is practically insoluble in water, slightly soluble in
History
Zafirlukast was the first cysteinyl leukotriene receptor antagonist approved in the United States.[10] Zafirlukast was approved in 1996.[10]
Society and culture
Economics
While preliminary evidence suggests that zafirlukast may reduce healthcare costs, the cost-effectiveness of using zafirlukast has not been established.[21]
Brand names
| A | Accolate, Accoleit, Aeronix |
| B | Benalucost |
| C | |
| D | |
| E | |
| F | Freesy |
| G | |
| H | |
| I | |
| J | |
| K | |
| L | |
| M | Monokast |
| N | |
| O | Olmoran |
| P | |
| Q | |
| R | |
| S | |
| T | |
| U | |
| V | Ventair |
| W | |
| X | |
| Y | |
| Z | Zafnil, Zalukast, Zukast |
Research
Mechanism of action
There is some research to suggest that zafirlukast actually acts as a partial inverse agonist at the CysLT1 receptor, though zafirlukast is still classified as an antagonist at this receptor. The possible clinical significance of this effect, if true, is unknown.[17]
Other indications
There is some evidence that suggests that zafirlukast may be beneficial in the treatment of chronic urticaria (hives), whether due to a known cause such as cold-exposure or due to an unknown cause (idiopathic).
Veterinary use
Zafirlukast is sometimes used for the treatment of bronchial asthma in cats.[23]
References
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab "ACCOLATE (zafirlukast) Package Insert" (PDF). www.accessdata.fda.gov. AstraZeneca LP. Retrieved 29 November 2017.
- ^ PMID 20332717.
- ^ "Asthma in children". ClinicalKey. Elsevier BV. October 11, 2017.
- ^ "Asthma, Hay Fever and Eczema: How to Cope with Atopic Triad". Inside Children's Blog. 29 April 2016. Archived from the original on 7 January 2018. Retrieved 7 January 2018.
- ^ )
- ^ ISBN 978-0-323-04289-5.
- ^ "Drug Safety Information for Healthcare Professionals – Updated Information on Leukotriene Inhibitors: Montelukast (marketed as Singulair), Zafirlukast (marketed as Accolate), and Zileuton (marketed as Zyflo and Zyflo CR)". Center for Drug Evaluation and Research. U.S. Food and Drug Administration. Archived from the original on November 14, 2017. Retrieved 6 December 2017.
- ^ )
- ^ "Accolate (zafirlukast) – Labeling Changes". www.medscape.com. WebMD LLC. Retrieved 29 November 2017.
- ^ S2CID 8124340.
- PMID 17767356.
- ^ "Medical Definition of Bronchoconstriction". www.merriam-webster.com. Merriam-Webster, Incorporated. Retrieved 29 November 2017.
- ^ "Medical Definition of Allergic Rhinitis". www.merriam-webster.com. Merriam-Webster, Incorporated. Retrieved 29 November 2017.
- ISBN 978-0-08-046884-6.
Cmax is the maximum concentration of the drug achieved in the plasma following dose administration and Tmax is the time at which Cmax is attained.
- ^ "Plasma albumin". TheFreeDictionary.com. Saunders. Retrieved 7 January 2018.
- PMID 9228664.
- ^ S2CID 24103280.
- ^ "Clinical Annotation for CYP2C9*1CYP2C9*13CYP2C9*3 related to zafirlukast". PharmGKB. Retrieved 29 November 2017.
- ^ "CYP2C9 Allele Functionality Table". PharmGKB. Retrieved 29 November 2017.
- .
- S2CID 249894596.
- ^ "Zafirlukast". Drugs.com. Retrieved 29 November 2017.
- ^ Byers CG, Dhupa N (June 2005). "Feline bronchial asthma: treatment" (PDF). Compend Contin Educ Pract Vet. 27 (6): 426–32.
External links
- "Zafirlukast". Drug Information Portal. U.S. National Library of Medicine.

