Zaytsev's rule
In
More generally, Zaytsev's rule predicts that in an elimination reaction the most substituted product will be the most stable, and therefore the most favored. The rule makes no generalizations about the stereochemistry of the newly formed alkene, but only the regiochemistry of the elimination reaction. While effective at predicting the favored product for many elimination reactions, Zaytsev's rule is subject to many exceptions. Many of them include exceptions under Hofmann product (analogous to Zaytsev product). These include compounds having quaternary nitrogen and leaving groups like NR3+, SO3H, etc. In these eliminations the Hofmann product is preferred. In case the leaving group is halogens, except fluorine; others give the Zaytsev product.[clarification needed]
History

Alexander Zaytsev first published his observations regarding the products of elimination reactions in Justus Liebigs Annalen der Chemie in 1875.[2][3] Although the paper contained some original research done by Zaytsev's students, it was largely a literature review and drew heavily upon previously published work.[4] In it, Zaytsev proposed a purely empirical rule for predicting the favored regiochemistry in the dehydrohalogenation of alkyl iodides, though it turns out that the rule is applicable to a variety of other elimination reactions as well. While Zaytsev's paper was well referenced throughout the 20th century, it was not until the 1960s that textbooks began using the term "Zaytsev's rule".[3]
Zaytsev was not the first chemist to publish the rule that now bears his name. Aleksandr Nikolaevich Popov published an empirical rule similar to Zaytsev's in 1872,[5] and presented his findings at the University of Kazan in 1873. Zaytsev had cited Popov's 1872 paper in previous work and worked at the University of Kazan, and was thus probably aware of Popov's proposed rule. In spite of this, Zaytsev's 1875 Liebigs Annalen paper makes no mention of Popov's work.[3][4]
Any discussion of Zaytsev's rule would be incomplete without mentioning
Thermodynamic considerations
The
The increase in stability associated with additional substitutions is the result of several factors.
Steric effects
In E2 elimination reactions, a base abstracts a proton that is beta to a leaving group, such as a halide. The removal of the proton and the loss of the leaving group occur in a single, concerted step to form a new double bond. When a small, unhindered base – such as sodium hydroxide, sodium methoxide, or sodium ethoxide – is used for an E2 elimination, the Zaytsev product is typically favored over the least substituted alkene, known as the Hofmann product. For example, treating 2-Bromo-2-methyl butane with sodium ethoxide in ethanol produces the Zaytsev product with moderate selectivity.[9]
Due to
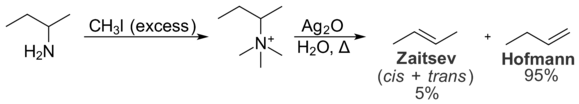
Steric interactions within the substrate also prevent the formation of the Zaytsev product. These intramolecular interactions are relevant to the distribution of products in the Hofmann elimination reaction, which converts amines to alkenes. In the Hofmann elimination, treatment of a quaternary ammonium iodide salt with silver oxide produces hydroxide ions, which act as a base and eliminate the tertiary amine to give an alkene.[11]
In the Hofmann elimination, the least substituted alkene is typically favored due to intramolecular steric interactions. The quaternary ammonium group is large, and interactions with alkyl groups on the rest of the molecule are undesirable. As a result, the conformation necessary for the formation of the Zaytsev product is less energetically favorable than the conformation required for the formation of the Hofmann product. As a result, the Hofmann product is formed preferentially. The
Stereochemistry
In some cases, the stereochemistry of the starting material can prevent the formation of the Zaytsev product. For example, when menthyl chloride is treated with sodium ethoxide, the Hofmann product is formed exclusively,[13] but in very low yield:[14]
This result is due to the stereochemistry of the starting material. E2 eliminations require
Formation of the Zaytsev product requires elimination at the 2-position, but the
See also
- Markovnikov's rule
- Hofmann elimination
- Cope elimination
References
- ISBN 978-0136000921.
- .
- ^ a b c d Lewis, D. E. (1995). "Alexander Mikhailovich Zaytsev (1841–1910) Markovnikov's Conservative Contemporary" (PDF). Bulletin for the History of Chemistry. 17: 21–30 (27).
- ^ PMID 21449203.
- .
- ^ Wade, pp. 292–294.
- ^ Wade, p. 293.
- ^ Wade, p. 301.
- ^ Wade, p. 302.
- ^ Wade, pp. 898–901.
- ^ Wade, p. 903.
- ^ Lehman 2009, pp. 183–184
- .
Bibliography
- Wade, L. G. (2010). Organic Chemistry (7th ed.). Upper Saddle River, NJ: Pearson Education. ISBN 978-0321592316.
External links
- Online course of chemistry
- [1] English Translation of 1875 German article on 'The order of addition and of elimination of hydrogen and iodine in organic compounds' by Alexander Zaytsev.