Zinc
 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Zinc | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Appearance | silver-gray | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard atomic weight Ar°(Zn) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Zinc in the periodic table | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of vaporization | 115 kJ/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar heat capacity | 25.470 J/(mol·K) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Vapor pressure
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Rasaratna Samuccaya (1300) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of zinc | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Zinc is a chemical element; it has symbol Zn and atomic number 30. It is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodic table. In some respects, it is chemically similar to magnesium: both elements exhibit only one normal oxidation state (+2), and the Zn2+ and Mg2+ ions are of similar size.[note 1] Zinc is the 24th most abundant element in Earth's crust and has five stable isotopes. The most common zinc ore is sphalerite (zinc blende), a zinc sulfide mineral. The largest workable lodes are in Australia, Asia, and the United States. Zinc is refined by froth flotation of the ore, roasting, and final extraction using electricity (electrowinning).
Zinc is an essential trace element for humans,[6][7][8] animals,[9] plants[10] and for microorganisms[11] and is necessary for prenatal and postnatal development.[12] It is the second most abundant trace metal in humans after iron, and the only metal which appears in all enzyme classes.[10][8] It is also an essential nutrient element for coral growth as it is an important cofactor for many enzymes.[13]
The element was probably named by the alchemist
Characteristics
Physical properties
Zinc is a bluish-white, lustrous,
Many alloys contain zinc, including brass. Other metals long known to form binary alloys with zinc are aluminium, antimony, bismuth, gold, iron, lead, mercury, silver, tin, magnesium, cobalt, nickel, tellurium, and sodium.[28] Although neither zinc nor zirconium is ferromagnetic, their alloy, ZrZn
2, exhibits ferromagnetism below 35 K.[23]
Occurrence
Zinc makes up about 75
Other source minerals for zinc include smithsonite (zinc carbonate), hemimorphite (zinc silicate), wurtzite (another zinc sulfide), and sometimes hydrozincite (basic zinc carbonate).[32] With the exception of wurtzite, all these other minerals were formed by weathering of the primordial zinc sulfides.[31]
Identified world zinc resources total about 1.9–2.8 billion tonnes.[33][34] Large deposits are in Australia, Canada and the United States, with the largest reserves in Iran.[31][35][36] The most recent estimate of reserve base for zinc (meets specified minimum physical criteria related to current mining and production practices) was made in 2009 and calculated to be roughly 480 Mt.[37] Zinc reserves, on the other hand, are geologically identified ore bodies whose suitability for recovery is economically based (location, grade, quality, and quantity) at the time of determination. Since exploration and mine development is an ongoing process, the amount of zinc reserves is not a fixed number and sustainability of zinc ore supplies cannot be judged by simply extrapolating the combined mine life of today's zinc mines. This concept is well supported by data from the United States Geological Survey (USGS), which illustrates that although refined zinc production increased 80% between 1990 and 2010, the reserve lifetime for zinc has remained unchanged. About 346 million tonnes have been extracted throughout history to 2002, and scholars have estimated that about 109–305 million tonnes are in use.[38][39][40]

Isotopes
Five stable isotopes of zinc occur in nature, with 64Zn being the most abundant isotope (49.17% natural abundance).[41][42] The other isotopes found in nature are 66
Zn (27.73%), 67
Zn (4.04%), 68
Zn (18.45%), and 70
Zn (0.61%).[42]
Several dozen
Zn has three excited metastable states and 73
Zn has two.[43] The isotopes 65
Zn, 71
Zn, 77
Zn and 78
Zn each have only one excited metastable state.[41]
The most common
The most common decay mode of a radioisotope of zinc with mass number higher than 66 is beta decay (β−), which produces an isotope of gallium.[41]
- n
30Zn
→ n
31Ga
+
ν
e
Compounds and chemistry
Reactivity
Zinc has an electron configuration of [Ar]3d104s2 and is a member of the group 12 of the periodic table. It is a moderately reactive metal and strong reducing agent.[44] The surface of the pure metal tarnishes quickly, eventually forming a protective passivating layer of the basic zinc carbonate, Zn
5(OH)
6(CO3)
2, by reaction with atmospheric carbon dioxide.[45]
Zinc burns in air with a bright bluish-green flame, giving off fumes of zinc oxide.[46] Zinc reacts readily with acids, alkalis and other non-metals.[47] Extremely pure zinc reacts only slowly at room temperature with acids.[46] Strong acids, such as hydrochloric or sulfuric acid, can remove the passivating layer and the subsequent reaction with the acid releases hydrogen gas.[46]
The chemistry of zinc is dominated by the +2 oxidation state. When compounds in this oxidation state are formed, the outer shell s electrons are lost, yielding a bare zinc ion with the electronic configuration [Ar]3d10.[48] In aqueous solution an octahedral complex, [Zn(H
2O)6]2+
is the predominant species.[49] The volatilization of zinc in combination with zinc chloride at temperatures above 285 °C indicates the formation of Zn
2Cl
2, a zinc compound with a +1 oxidation state.[46] No compounds of zinc in positive oxidation states other than +1 or +2 are known.[50] Calculations indicate that a zinc compound with the oxidation state of +4 is unlikely to exist.[51] Zn(III) is predicted to exist in the presence of strongly electronegative trianions;[52] however, there exists some doubt around this possibility.[53] But in 2021 another compound was reported with more evidence that had the oxidation state of +3 with the formula ZnBeB11(CN)12.[54]
Zinc chemistry is similar to the chemistry of the late first-row transition metals,
Zinc(I) compounds
Zinc(I) compounds are very rare. The [Zn2]2+ ion is implicated by the formation of a yellow diamagnetic glass by dissolving metallic zinc in molten ZnCl2.[57] The [Zn2]2+ core would be analogous to the [Hg2]2+ cation present in mercury(I) compounds. The diamagnetic nature of the ion confirms its dimeric structure. The first zinc(I) compound containing the Zn–Zn bond, (η5-C5Me5)2Zn2.
Zinc(II) compounds

3CO
2)
2

3N
2, Zn
3P
2, Zn
3As
2 and Zn
3Sb
2),[59][60] the peroxide (ZnO
2), the hydride (ZnH
2), and the carbide (ZnC
2) are also known.[61] Of the four halides, ZnF
2 has the most ionic character, while the others (ZnCl
2, ZnBr
2, and ZnI
2) have relatively low melting points and are considered to have more covalent character.[62]
In weak basic solutions containing Zn2+
ions, the hydroxide
).[46] The nitrate Zn(NO3)
2, chlorate Zn(ClO3)
2, sulfate ZnSO
4, phosphate Zn
3(PO4)
2, molybdate ZnMoO
4, cyanide Zn(CN)
2, arsenite Zn(AsO2)
2, arsenate Zn(AsO4)
2·8H
2O and the chromate ZnCrO
4 (one of the few colored zinc compounds) are a few examples of other common inorganic compounds of zinc.[63][64]
2H5)
2Zn) is a reagent in synthetic chemistry. It was first reported in 1848 from the reaction of zinc and ethyl iodide, and was the first compound known to contain a metal–carbon sigma bond.[65]
Test for zinc
Cobalticyanide paper (Rinnmann's test for Zn) can be used as a chemical indicator for zinc. 4 g of K3Co(CN)6 and 1 g of KClO3 is dissolved on 100 ml of water. Paper is dipped in the solution and dried at 100 °C. One drop of the sample is dropped onto the dry paper and heated. A green disc indicates the presence of zinc.[66]
History
Ancient use
Various isolated examples of the use of impure zinc in ancient times have been discovered. Zinc ores were used to make the zinc–copper alloy brass thousands of years prior to the discovery of zinc as a separate element. Judean brass from the 14th to 10th centuries BC contains 23% zinc.[18]
Knowledge of how to produce brass spread to Ancient Greece by the 7th century BC, but few varieties were made.[19] Ornaments made of alloys containing 80–90% zinc, with lead, iron, antimony, and other metals making up the remainder, have been found that are 2,500 years old.[30] A possibly prehistoric statuette containing 87.5% zinc was found in a Dacian archaeological site.[67]
Strabo writing in the 1st century BC (but quoting a now lost work of the 4th century BC historian Theopompus) mentions "drops of false silver" which when mixed with copper make brass. This may refer to small quantities of zinc that is a by-product of smelting sulfide ores.[68] Zinc in such remnants in smelting ovens was usually discarded as it was thought to be worthless.[69]
The manufacture of brass was known to the Romans by about 30 BC.[70] They made brass by heating powdered calamine (zinc silicate or carbonate), charcoal and copper together in a crucible.[70] The resulting calamine brass was then either cast or hammered into shape for use in weaponry.[71] Some coins struck by Romans in the Christian era are made of what is probably calamine brass.[72]

The oldest known pills were made of the zinc carbonates hydrozincite and smithsonite. The pills were used for sore eyes and were found aboard the Roman ship Relitto del Pozzino, wrecked in 140 BC.[73][74]
The
The
Early studies and naming
Zinc was distinctly recognized as a metal under the designation of Yasada or Jasada in the medical Lexicon ascribed to the Hindu king Madanapala (of Taka dynasty) and written about the year 1374.[80] Smelting and extraction of impure zinc by reducing calamine with wool and other organic substances was accomplished in the 13th century in India.[23][81] The Chinese did not learn of the technique until the 17th century.[81]

Alchemists burned zinc metal in air and collected the resulting zinc oxide on a condenser. Some alchemists called this zinc oxide lana philosophica, Latin for "philosopher's wool", because it collected in wooly tufts, whereas others thought it looked like white snow and named it nix album.[82]
The name of the metal was probably first documented by Paracelsus, a Swiss-born German alchemist, who referred to the metal as "zincum" or "zinken" in his book Liber Mineralium II, in the 16th century.[81][83] The word is probably derived from the German zinke, and supposedly meant "tooth-like, pointed or jagged" (metallic zinc crystals have a needle-like appearance).[84] Zink could also imply "tin-like" because of its relation to German zinn meaning tin.[85] Yet another possibility is that the word is derived from the Persian word سنگ seng meaning stone.[86] The metal was also called Indian tin, tutanego, calamine, and spinter.[30]
German metallurgist Andreas Libavius received a quantity of what he called "calay" (from the Malay or Hindi word for tin) originating from Malabar off a cargo ship captured from the Portuguese in the year 1596.[87] Libavius described the properties of the sample, which may have been zinc. Zinc was regularly imported to Europe from the Orient in the 17th and early 18th centuries,[81] but was at times very expensive.[note 2]
Isolation

Metallic zinc was isolated in India by 1300 AD.
Flemish
In 1738 in Great Britain, William Champion patented a process to extract zinc from calamine in a vertical retort-style smelter.[94] His technique resembled that used at Zawar zinc mines in Rajasthan, but no evidence suggests he visited the Orient.[91] Champion's process was used through 1851.[81]
German chemist Andreas Marggraf normally gets credit for isolating pure metallic zinc in the West, even though Swedish chemist Anton von Swab had distilled zinc from calamine four years previously.[81] In his 1746 experiment, Marggraf heated a mixture of calamine and charcoal in a closed vessel without copper to obtain a metal.[95][69] This procedure became commercially practical by 1752.[96]
Later work

William Champion's brother, John, patented a process in 1758 for
Galvani's friend, Alessandro Volta, continued researching the effect and invented the Voltaic pile in 1800.[98] Volta's pile consisted of a stack of simplified galvanic cells, each being one plate of copper and one of zinc connected by an electrolyte. By stacking these units in series, the Voltaic pile (or "battery") as a whole had a higher voltage, which could be used more easily than single cells. Electricity is produced because the Volta potential between the two metal plates makes electrons flow from the zinc to the copper and corrode the zinc.[98]
The non-magnetic character of zinc and its lack of color in solution delayed discovery of its importance to biochemistry and nutrition.[100] This changed in 1940 when carbonic anhydrase, an enzyme that scrubs carbon dioxide from blood, was shown to have zinc in its active site.[100] The digestive enzyme carboxypeptidase became the second known zinc-containing enzyme in 1955.[100]
Production
Mining and processing
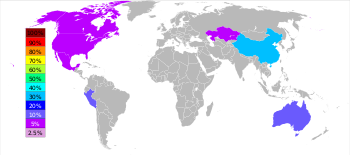
| Rank | Country | Tonnes |
|---|---|---|
| 1 | China | 4,210,000 |
| 2 | Peru | 1,400,000 |
| 3 | Australia | 1,330,000 |
| 4 | United States | 753,000 |
| 5 | India | 720,000 |
| 6 | Mexico | 677,000 |

27°57′17″S 016°46′00″E / 27.95472°S 16.76667°E

27°49′09″S 016°36′28″E / 27.81917°S 16.60778°E
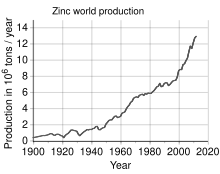
Zinc is the fourth most common metal in use, trailing only iron, aluminium, and copper with an annual production of about 13 million tonnes.[33] The world's largest zinc producer is Nyrstar, a merger of the Australian OZ Minerals and the Belgian Umicore.[102] About 70% of the world's zinc originates from mining, while the remaining 30% comes from recycling secondary zinc.[103]
Commercially pure zinc is known as Special High Grade, often abbreviated SHG, and is 99.995% pure.[104]
Worldwide, 95% of new zinc is mined from sulfidic ore deposits, in which sphalerite (ZnS) is nearly always mixed with the sulfides of copper, lead and iron.[105]: 6 Zinc mines are scattered throughout the world, with the main areas being China, Australia, and Peru. China produced 38% of the global zinc output in 2014.[33]
Zinc metal is produced using
2.[106]
Roasting converts the zinc sulfide concentrate to zinc oxide:[105]
The sulfur dioxide is used for the production of sulfuric acid, which is necessary for the leaching process. If deposits of
For further processing two basic methods are used: pyrometallurgy or electrowinning. Pyrometallurgy reduces zinc oxide with carbon or carbon monoxide at 950 °C (1,740 °F) into the metal, which is distilled as zinc vapor to separate it from other metals, which are not volatile at those temperatures.[108] The zinc vapor is collected in a condenser.[105] The equations below describe this process:[105]
In electrowinning, zinc is leached from the ore concentrate by sulfuric acid and impurities are precipitated:[109]
Finally, the zinc is reduced by electrolysis.[105]
The sulfuric acid is regenerated and recycled to the leaching step.
When galvanised feedstock is fed to an electric arc furnace, the zinc is recovered from the dust by a number of processes, predominantly the Waelz process (90% as of 2014).[110]
Environmental impact
Refinement of sulfidic zinc ores produces large volumes of sulfur dioxide and
Zinc in rivers flowing through industrial and mining areas can be as high as 20 ppm.[112] Effective sewage treatment greatly reduces this; treatment along the Rhine, for example, has decreased zinc levels to 50 ppb.[112] Concentrations of zinc as low as 2 ppm adversely affects the amount of oxygen that fish can carry in their blood.[113]
Applications
Major applications of zinc include, with percentages given for the US[117]
- Galvanizing (55%)
- Brass and bronze (16%)
- Other alloys (21%)
- Miscellaneous (8%)
Anti-corrosion and batteries

Zinc is most commonly used as an anti-corrosion agent,[118] and galvanization (coating of iron or steel) is the most familiar form. In 2009 in the United States, 55% or 893,000 tons of the zinc metal was used for galvanization.[117]
Zinc is more reactive than iron or steel and thus will attract almost all local oxidation until it completely corrodes away.
The relative reactivity of zinc and its ability to attract oxidation to itself makes it an efficient
With a
Alloys
A widely used zinc alloy is brass, in which copper is alloyed with anywhere from 3% to 45% zinc, depending upon the type of brass.

Other widely used zinc alloys include nickel silver, typewriter metal, soft and aluminium solder, and commercial bronze.[23] Zinc is also used in contemporary pipe organs as a substitute for the traditional lead/tin alloy in pipes.[129] Alloys of 85–88% zinc, 4–10% copper, and 2–8% aluminium find limited use in certain types of machine bearings. Zinc has been the primary metal in American one cent coins (pennies) since 1982.[130] The zinc core is coated with a thin layer of copper to give the appearance of a copper coin. In 1994, 33,200 tonnes (36,600 short tons) of zinc were used to produce 13.6 billion pennies in the United States.[131]
Alloys of zinc with small amounts of copper, aluminium, and magnesium are useful in die casting as well as spin casting, especially in the automotive, electrical, and hardware industries.[23] These alloys are marketed under the name Zamak.[132] An example of this is zinc aluminium. The low melting point together with the low viscosity of the alloy makes possible the production of small and intricate shapes. The low working temperature leads to rapid cooling of the cast products and fast production for assembly.[23][133] Another alloy, marketed under the brand name Prestal, contains 78% zinc and 22% aluminium, and is reported to be nearly as strong as steel but as malleable as plastic.[23][134] This superplasticity of the alloy allows it to be molded using die casts made of ceramics and cement.[23]
Similar alloys with the addition of a small amount of lead can be cold-rolled into sheets. An alloy of 96% zinc and 4% aluminium is used to make stamping dies for low production run applications for which ferrous metal dies would be too expensive.[135] For building facades, roofing, and other applications for sheet metal formed by deep drawing, roll forming, or bending, zinc alloys with titanium and copper are used.[136] Unalloyed zinc is too brittle for these manufacturing processes.[136]
As a dense, inexpensive, easily worked material, zinc is used as a lead replacement. In the wake of lead concerns, zinc appears in weights for various applications ranging from fishing[137] to tire balances and flywheels.[138]
Cadmium zinc telluride (CZT) is a semiconductive alloy that can be divided into an array of small sensing devices.[139] These devices are similar to an integrated circuit and can detect the energy of incoming gamma ray photons.[139] When behind an absorbing mask, the CZT sensor array can determine the direction of the rays.[139]
Other industrial uses

Roughly one quarter of all zinc output in the United States in 2009 was consumed in zinc compounds;
Zinc powder is sometimes used as a propellant in model rockets.[148] When a compressed mixture of 70% zinc and 30% sulfur powder is ignited there is a violent chemical reaction.[148] This produces zinc sulfide, together with large amounts of hot gas, heat, and light.[148]
Zinc sheet metal is used as a durable covering for roofs, walls, and countertops, the last often seen in
64
Zn, the most abundant isotope of zinc, is very susceptible to neutron activation, being transmuted into the highly radioactive 65
Zn, which has a half-life of 244 days and produces intense gamma radiation. Because of this, zinc oxide used in nuclear reactors as an anti-corrosion agent is depleted of 64
Zn before use, this is called depleted zinc oxide. For the same reason, zinc has been proposed as a salting material for nuclear weapons (cobalt is another, better-known salting material).[153] A jacket of isotopically enriched 64
Zn would be irradiated by the intense high-energy neutron flux from an exploding thermonuclear weapon, forming a large amount of 65
Zn significantly increasing the radioactivity of the weapon's fallout.[153] Such a weapon is not known to have ever been built, tested, or used.[153]
65
Zn is used as a
Zinc dithiocarbamate complexes are used as agricultural fungicides; these include Zineb, Metiram, Propineb and Ziram.[155] Zinc naphthenate is used as wood preservative.[156] Zinc in the form of ZDDP, is used as an anti-wear additive for metal parts in engine oil.[157]
Organic chemistry

- The Frankland-Duppa Reaction in which an alkyl halide R'X, zinc and hydrochloric acid to form α-hydroxycarboxylic esters RR'COHCOOR[163][164]
- Organozincs have similar reactivity to organobromineprecursors.
Zinc has found many uses in catalysis in organic synthesis including
Dietary supplement


Zinc, a vital trace mineral, is not stored in the body in large quantities, necessitating regular dietary intake for optimal health. Regular intake is particularly crucial given zinc's extensive involvement in human health, including its roles in cellular metabolism (zinc is integral to the function of over 300 enzymes in the human body), immune function, protein synthesis, DNA synthesis, and cell division.[166]
In most single-tablet, over-the-counter, daily
Generally, zinc supplement is recommended where there is high risk of zinc deficiency (such as low and middle income countries) as a preventive measure.[169] Although zinc sulfate is a commonly used zinc form, zinc citrate, gluconate and picolinate may be valid options as well. These forms are better absorbed than zinc oxide.[170]
Gastroenteritis
Zinc is an inexpensive and effective part of treatment of diarrhea among children in the developing world. Zinc becomes depleted in the body during diarrhea and replenishing zinc with a 10- to 14-day course of treatment can reduce the duration and severity of diarrheal episodes and may also prevent future episodes for as long as three months.[171] Gastroenteritis is strongly attenuated by ingestion of zinc, possibly by direct antimicrobial action of the ions in the gastrointestinal tract, or by the absorption of the zinc and re-release from immune cells (all granulocytes secrete zinc), or both.[172][173]
Common cold
Zinc supplements (frequently
Weight gain
Zinc deficiency may lead to loss of appetite.[177] The use of zinc in the treatment of anorexia has been advocated since 1979. At least 15 clinical trials have shown that zinc improved weight gain in anorexia. A 1994 trial showed that zinc doubled the rate of body mass increase in the treatment of anorexia nervosa. Deficiency of other nutrients such as tyrosine, tryptophan and thiamine could contribute to this phenomenon of "malnutrition-induced malnutrition".[178] A meta-analysis of 33 prospective intervention trials regarding zinc supplementation and its effects on the growth of children in many countries showed that zinc supplementation alone had a statistically significant effect on linear growth and body weight gain, indicating that other deficiencies that may have been present were not responsible for growth retardation.[179]
Other
A 2023 Cochrane review stated that people taking zinc supplements may be less likely to progress to
Topical use
Chelated zinc is used in toothpastes and mouthwashes to prevent
Zinc pyrithione is widely included in shampoos to prevent dandruff.[186]
Topical zinc has also been shown to effectively treat, as well as prolong remission in genital herpes.[187]
Biological role
Zinc is an essential trace element for humans[188][6][7][8] and other animals,[9] for plants[10] and for microorganisms.[11] Zinc is required for the function of over 300 enzymes and 1000 transcription factors,[166][8] and is stored and transferred in metallothioneins.[189][190] It is the second most abundant trace metal in humans after iron and it is the only metal which appears in all enzyme classes.[10][8]
In proteins, zinc ions are often coordinated to the amino acid side chains of aspartic acid, glutamic acid, cysteine and histidine. The theoretical and computational description of this zinc binding in proteins (as well as that of other transition metals) is difficult.[191]
Roughly 2–4 grams of zinc
Zinc homeostasis of the body is mainly controlled by the intestine. Here,
In humans, the biological roles of zinc are ubiquitous.[12][7] It interacts with "a wide range of organic ligands",[12] and has roles in the metabolism of RNA and DNA, signal transduction, and gene expression. It also regulates apoptosis. A review from 2015 indicated that about 10% of human proteins (~3000) bind zinc,[197] in addition to hundreds more that transport and traffic zinc; a similar in silico study in the plant Arabidopsis thaliana found 2367 zinc-related proteins.[10]
In the
Enzymes

Zinc is an efficient
2) regulation and digestion of proteins, respectively.[204]
In vertebrate blood, carbonic anhydrase converts CO
2 into bicarbonate and the same enzyme transforms the bicarbonate back into CO
2 for exhalation through the lungs.
Carboxypeptidase cleaves peptide linkages during digestion of proteins. A
Signalling
Zinc has been recognized as a messenger, able to activate signalling pathways. Many of these pathways provide the driving force in aberrant cancer growth. They can be targeted through ZIP transporters.[209]
Other proteins
Zinc serves a purely structural role in
ions in a zinc finger helps maintain the finger's structure by coordinately binding to four amino acids in the transcription factor.[206]
In blood plasma, zinc is bound to and transported by albumin (60%, low-affinity) and transferrin (10%).[192] Because transferrin also transports iron, excessive iron reduces zinc absorption, and vice versa. A similar antagonism exists with copper.[211] The concentration of zinc in blood plasma stays relatively constant regardless of zinc intake.[202] Cells in the salivary gland, prostate, immune system, and intestine use zinc signaling to communicate with other cells.[212]
Zinc may be held in metallothionein reserves within microorganisms or in the intestines or liver of animals.[213] Metallothionein in intestinal cells is capable of adjusting absorption of zinc by 15–40%.[214] However, inadequate or excessive zinc intake can be harmful; excess zinc particularly impairs copper absorption because metallothionein absorbs both metals.[215]
The human
Nutrition
Dietary recommendations
The
The
For U.S. food and dietary supplement labeling purposes the amount in a serving is expressed as a percent of Daily Value (%DV). For zinc labeling purposes 100% of the Daily Value was 15 mg, but on May 27, 2016, it was revised to 11 mg.[222][223] A table of the old and new adult daily values is provided at Reference Daily Intake.
Dietary intake
Animal products such as meat, fish, shellfish, fowl, eggs, and dairy contain zinc. The concentration of zinc in plants varies with the level in the soil. With adequate zinc in the soil, the food plants that contain the most zinc are wheat (germ and bran) and various seeds, including sesame, poppy, alfalfa, celery, and mustard.[224] Zinc is also found in beans, nuts, almonds, whole grains, pumpkin seeds, sunflower seeds, and blackcurrant.[225]
Other sources include fortified food and dietary supplements in various forms. A 1998 review concluded that zinc oxide, one of the most common supplements in the United States, and zinc carbonate are nearly insoluble and poorly absorbed in the body.[226] This review cited studies that found lower plasma zinc concentrations in the subjects who consumed zinc oxide and zinc carbonate than in those who took zinc acetate and sulfate salts.[226] For fortification, however, a 2003 review recommended cereals (containing zinc oxide) as a cheap, stable source that is as easily absorbed as the more expensive forms.[227] A 2005 study found that various compounds of zinc, including oxide and sulfate, did not show statistically significant differences in absorption when added as fortificants to maize tortillas.[228]
Deficiency
Nearly two billion people in the developing world are deficient in zinc. Groups at risk include children in developing countries and elderly with chronic illnesses.[14] In children, it causes an increase in infection and diarrhea and contributes to the death of about 800,000 children worldwide per year.[12] The World Health Organization advocates zinc supplementation for severe malnutrition and diarrhea.[229] Zinc supplements help prevent disease and reduce mortality, especially among children with low birth weight or stunted growth.[229] However, zinc supplements should not be administered alone, because many in the developing world have several deficiencies, and zinc interacts with other micronutrients.[230] While zinc deficiency is usually due to insufficient dietary intake, it can be associated with malabsorption, acrodermatitis enteropathica, chronic liver disease, chronic renal disease, sickle cell disease, diabetes, malignancy, and other chronic illnesses.[14]
In the United States, a federal survey of food consumption determined that for women and men over the age of 19, average consumption was 9.7 and 14.2 mg/day, respectively. For women, 17% consumed less than the EAR, for men 11%. The percentages below EAR increased with age.[231] The most recent published update of the survey (NHANES 2013–2014) reported lower averages – 9.3 and 13.2 mg/day – again with intake decreasing with age.[232]
Symptoms of mild zinc deficiency are diverse.
Despite some concerns,
Soil remediation
Species of Calluna, Erica and Vaccinium can grow in zinc-metalliferous soils, because translocation of toxic ions is prevented by the action of ericoid mycorrhizal fungi.[237]
Agriculture
Zinc deficiency appears to be the most common micronutrient deficiency in crop plants; it is particularly common in high-pH soils.[238] Zinc-deficient soil is cultivated in the cropland of about half of Turkey and India, a third of China, and most of Western Australia. Substantial responses to zinc fertilization have been reported in these areas.[10] Plants that grow in soils that are zinc-deficient are more susceptible to disease. Zinc is added to the soil primarily through the weathering of rocks, but humans have added zinc through fossil fuel combustion, mine waste, phosphate fertilizers, pesticide (zinc phosphide), limestone, manure, sewage sludge, and particles from galvanized surfaces. Excess zinc is toxic to plants, although zinc toxicity is far less widespread.[10]
Precautions
Toxicity
Although zinc is an essential requirement for good health, excess zinc can be harmful. Excessive absorption of zinc suppresses copper and iron absorption.[215] The free zinc ion in solution is highly toxic to plants, invertebrates, and even vertebrate fish.[239] The Free Ion Activity Model is well-established in the literature, and shows that just micromolar amounts of the free ion kills some organisms. A recent example showed 6 micromolar killing 93% of all Daphnia in water.[240]
The free zinc ion is a powerful
Evidence shows that people taking 100–300 mg of zinc daily may suffer induced
The U.S. Food and Drug Administration (FDA) states that zinc damages nerve receptors in the nose, causing anosmia. Reports of anosmia were also observed in the 1930s when zinc preparations were used in a failed attempt to prevent polio infections.[244] On June 16, 2009, the FDA ordered removal of zinc-based intranasal cold products from store shelves. The FDA said the loss of smell can be life-threatening because people with impaired smell cannot detect leaking gas or smoke, and cannot tell if food has spoiled before they eat it.[245]
Recent research suggests that the topical antimicrobial zinc pyrithione is a potent
Poisoning
In 1982, the
Pennies and other small coins are sometimes ingested by dogs, requiring veterinary removal of the foreign objects. The zinc content of some coins can cause zinc toxicity, commonly fatal in dogs through severe
See also
- List of countries by zinc production
- Spelter
- Wet storage stain
- Zinc alloy electroplating
- Metal fume fever
- Piotr Steinkeller
Notes
- ^ The elements are from different metal groups. See periodic table.
- ^ An East India Company ship carrying a cargo of nearly pure zinc metal from the Orient sank off the coast Sweden in 1745.(Emsley 2001, p. 502)
- ^ Electric current will naturally flow between zinc and steel but in some circumstances inert anodes are used with an external DC source.
Citations
- ^ "Standard Atomic Weights: Zinc". CIAAW. 2007.
- ISSN 1365-3075.
- ISBN 978-1-62708-155-9.
- ISBN 0-8493-0464-4.
- ^ .
- ^ PMID 24470098.
- ^ S2CID 21141511.
- ^ PMID 29113075.
Zinc is the second most abundant trace metal in the human body, and is essential for many biological processes. ... The trace metal zinc is an essential cofactor for more than 300 enzymes and 1000 transcription factors [16]. ... In the central nervous system, zinc is the second most abundant trace metal and is involved in many processes. In addition to its role in enzymatic activity, it also plays a major role in cell signaling and modulation of neuronal activity.
- ^ PMID 18385818.
- ^ PMID 17286818.
- ^ PMID 6338570.
- ^ PMID 17374687.
- from the original on November 22, 2022. Retrieved November 22, 2022.
- ^ PMID 12595353.
- PMID 23595681.
- ^ Anglia, University of East. "Zinc vital to evolution of complex life in polar oceans". phys.org. Archived from the original on September 3, 2023. Retrieved September 3, 2023.
- ISBN 978-1-904982-19-7. Archived(PDF) from the original on September 24, 2015 – via Papers and Lectures Online.
- ^ a b Greenwood & Earnshaw 1997, p. 1201
- ^ .
- ^ "Zinc – Royal Society Of Chemistry". Archived from the original on July 11, 2017.
- ^ "India Was the First to Smelt Zinc by Distillation Process". Infinityfoundation.com. Archived from the original on May 16, 2016. Retrieved April 25, 2014.
- doi:10.5334/aa.06112.
- ^ a b c d e f g h i j CRC 2006, p. 4–41
- ^ a b Heiserman 1992, p. 123
- ISBN 0-19-855370-6
- ^ Scoffern, John (1861). The Useful Metals and Their Alloys. Houlston and Wright. pp. 591–603. Archived from the original on March 17, 2024. Retrieved April 6, 2009.
- ^ a b "Zinc Metal Properties". American Galvanizers Association. 2008. Archived from the original on March 28, 2015. Retrieved April 7, 2015.
- ^ Ingalls, Walter Renton (1902). "Production and Properties of Zinc: A Treatise on the Occurrence and Distribution of Zinc Ore, the Commercial and Technical Conditions Affecting the Production of the Spelter, Its Chemical and Physical Properties and Uses in the Arts, Together with a Historical and Statistical Review of the Industry". The Engineering and Mining Journal: 142–6. Archived from the original on March 17, 2024. Retrieved September 27, 2020.
- from the original on June 3, 2023. Retrieved November 11, 2021.
- ^ a b c d e Lehto 1968, p. 822
- ^ a b c Greenwood & Earnshaw 1997, p. 1202
- ^ a b c d Emsley 2001, p. 502
- ^ a b c d Sai Srujan, A.V (2021). "Mineral Commodity Summaries 2021: Zinc" (PDF). United States Geological Survey. Archived (PDF) from the original on June 3, 2023. Retrieved June 21, 2021.
- ^ Erickson, R. L. (1973). "Crustal Abundance of Elements, and Mineral Reserves and Resources". U.S. Geological Survey Professional Paper (820): 21–25.
- ^ "Country Partnership Strategy—Iran: 2011–12". ECO Trade and development bank. Archived from the original on October 26, 2011. Retrieved June 6, 2011.
- ^ "IRAN – a growing market with enormous potential". IMRG. July 5, 2010. Archived from the original on February 17, 2013. Retrieved March 3, 2010.
- ^ Tolcin, A. C. (2009). "Mineral Commodity Summaries 2009: Zinc" (PDF). United States Geological Survey. Archived (PDF) from the original on July 2, 2016. Retrieved August 4, 2016.
- PMID 16432205.
- PMID 18939524.
- .
- ^ a b c d e Alejandro A. Sonzogni (Database Manager), ed. (2008). "Chart of Nuclides". Upton (NY): National Nuclear Data Center, Brookhaven National Laboratory. Archived from the original on May 22, 2008. Retrieved September 13, 2008.
- ^ .
- ^ CRC 2006, pp. 8–29
- ISBN 978-0-8247-9213-8.
- ^ ISBN 978-3-11-007511-3.
- ^ Hinds, John Iredelle Dillard (1908). Inorganic Chemistry: With the Elements of Physical and Theoretical Chemistry (2nd ed.). New York: John Wiley & Sons. pp. 506–508. Archived from the original on March 17, 2024. Retrieved September 27, 2020.
- ISBN 978-1-84315-438-9. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ISBN 978-0-470-26293-1.
- ISBN 978-0-471-86739-5.
- from the original on June 4, 2023. Retrieved September 4, 2019.
- from the original on June 4, 2023. Retrieved August 27, 2021.
- from the original on June 4, 2023. Retrieved August 27, 2021.
- S2CID 237400349.
- ^ a b Greenwood & Earnshaw 1997, p. 1206
- ^ CRC 2006, pp. 12–11–12
- ISBN 978-0-13-175553-6.
- ^ "Zinc Sulfide". American Elements. Archived from the original on July 17, 2012. Retrieved February 3, 2009.
- ISBN 978-0-7172-2053-3. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ^ "Zinc Phosphide". American Elements. Archived from the original on July 17, 2012. Retrieved February 3, 2009.
- .
- ^ Greenwood & Earnshaw 1997, p. 1211
- ^ Rasmussen, J. K.; Heilmann, S. M. (1990). "In situ Cyanosilylation of Carbonyl Compounds: O-Trimethylsilyl-4-Methoxymandelonitrile". Organic Syntheses, Collected Volume. 7: 521. Archived from the original on September 30, 2007.
- ISBN 978-0-8493-8671-8.
- from the original on June 4, 2023. Retrieved September 27, 2020.
- ISBN 978-0-8493-0479-8.
- ^ Weeks 1933, p. 20
- ISBN 978-0-86159-124-4.
- ^ a b Weeks 1933, p. 21
- ^ a b c d e f Emsley 2001, p. 501
- ^ "How is zinc made?". How Products are Made. The Gale Group. 2002. Archived from the original on April 11, 2006. Retrieved February 21, 2009.
- ^ Chambers 1901, p. 799
- ^ "World's oldest pills treated sore eyes". New Scientist. January 7, 2013. Archived from the original on January 22, 2013. Retrieved February 5, 2013.
- PMID 23297212.
- ^ Rehren, Th. (1996). S. Demirci; et al. (eds.). A Roman zinc tablet from Bern, Switzerland: Reconstruction of the Manufacture. Archaeometry 94. The Proceedings of the 29th International Symposium on Archaeometry. pp. 35–45.
- OCLC 165833440.
- ISBN 978-0-86159-124-4.
- ^ ISBN 1-84265-001-7.
- ^ JSTOR 124653.
- ^ Ray, Prafulla Chandra (1903). A History of Hindu Chemistry from the Earliest Times to the Middle of the Sixteenth Century, A.D.: With Sanskrit Texts, Variants, Translation and Illustrations. Vol. 1 (2nd ed.). The Bengal Chemical & Pharmaceutical Works, Ltd. pp. 157–158. Archived from the original on March 17, 2024. Retrieved September 27, 2020. (public domain text)
- ^ a b c d e f g Habashi, Fathi. "Discovering the 8th Metal" (PDF). International Zinc Association (IZA). Archived from the original (PDF) on March 4, 2009. Retrieved December 13, 2008.
- ^ Arny, Henry Vinecome (1917). Principles of Pharmacy (2nd ed.). W. B. Saunders company. p. 483. Archived from the original on March 17, 2024. Retrieved September 27, 2020.
- ISBN 978-0-7661-3197-2.
- ISBN 978-3-527-20100-6.
- ISBN 978-1-59605-092-1. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ISBN 978-3-527-28792-5.
- ISBN 978-0-226-46734-4. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ^ Vaughan, L Brent (1897). "Zincography". The Junior Encyclopedia Britannica A Reference Library of General Knowledge Volume III P-Z. Chicago: E. G. Melven & Company.
- ^ Castellani, Michael. "Transition Metal Elements" (PDF). Archived (PDF) from the original on October 10, 2014. Retrieved October 14, 2014.
- ISBN 978-81-317-2791-1. Archivedfrom the original on April 14, 2016.
- ^ .
- JSTOR 124574.
- ^ Roberts, R. O. (1951). "Dr John Lane and the foundation of the non-ferrous metal industry in the Swansea valley". Gower (4). Gower Society: 19.
- ISBN 978-0-8493-9163-7. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ^ Marggraf (1746). "Experiences sur la maniere de tirer le Zinc de sa veritable miniere, c'est à dire, de la pierre calaminaire" [Experiments on a way of extracting zinc from its true mineral; i.e., the stone calamine]. Histoire de l'Académie Royale des Sciences et Belles-Lettres de Berlin (in French). 2: 49–57. Archived from the original on June 4, 2023. Retrieved October 16, 2020.
- ^ Heiserman 1992, p. 122
- ISBN 978-0-7614-1922-8.
- ^ ISBN 978-1-74020-085-1. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ^ a b "Galvanic Cell". The New International Encyclopaedia. Dodd, Mead and Company. 1903. p. 80. Archived from the original on March 17, 2024. Retrieved September 27, 2020.
- ^ a b c Cotton et al. 1999, p. 626
- ^ Jasinski, Stephen M. "Mineral Commodity Summaries 2007: Zinc" (PDF). United States Geological Survey. Archived (PDF) from the original on December 17, 2008. Retrieved November 25, 2008.
- ^ Attwood, James (February 13, 2006). "Zinifex, Umicore Combine to Form Top Zinc Maker". The Wall Street Journal. Archived from the original on January 26, 2017.
- ^ "Zinc Recycling". International Zinc Association. Archived from the original on October 21, 2011. Retrieved November 28, 2008.
- ^ "Special High Grade Zinc (SHG) 99.995%" (PDF). Nyrstar. 2008. Archived from the original (PDF) on March 4, 2009. Retrieved December 1, 2008.
- ^ ISBN 978-0-8247-8340-2. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ^ ISBN 978-82-519-1922-7.
- doi:10.2113/98.4.749.
- ISBN 978-0-8493-4433-6.
- ISBN 978-0-8493-6804-2.
- ^ Antrekowitsch, Jürgen; Steinlechner, Stefan; Unger, Alois; Rösler, Gernot; Pichler, Christoph; Rumpold, Rene (2014), "9. Zinc and Residue Recycling", in Worrell, Ernst; Reuter, Markus (eds.), Handbook of Recycling: State-of-the-art for Practitioners, Analysts, and Scientists
- ^ S2CID 129717791.
- ^ a b c d Emsley 2001, p. 504
- ISBN 978-0-87371-632-1. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ^ "Derwent Estuary – Water Quality Improvement Plan for Heavy Metals". Derwent Estuary Program. June 2007. Archived from the original on March 21, 2012. Retrieved July 11, 2009.
- ^ "The Zinc Works". TChange. Archived from the original on April 27, 2009. Retrieved July 11, 2009.
- from the original on October 14, 2023. Retrieved February 21, 2024.
- ^ a b c "Zinc: World Mine Production (zinc content of concentrate) by Country" (PDF). 2009 Minerals Yearbook: Zinc. Washington, D.C.: United States Geological Survey. February 2010. Archived (PDF) from the original on June 8, 2011. Retrieved June 6, 2001.
- ^ Greenwood & Earnshaw 1997, p. 1203
- ^ a b Stwertka 1998, p. 99
- ^ a b c d e f g Lehto 1968, p. 829
- ^ a b Emsley 2001, p. 503
- S2CID 135744939.
- ISBN 978-3-527-29469-5.
- .
- S2CID 106826667.
- ^ Whartman, Jonathan; Brown, Ian. "Zinc Air Battery-Battery Hybrid for Powering Electric Scooters and Electric Buses" (PDF). The 15th International Electric Vehicle Symposium. Archived from the original (PDF) on March 12, 2006. Retrieved October 8, 2008.
- OSTI 82465.
- .
- ISBN 978-0-415-94174-7. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ^ "Coin Specifications". United States Mint. Archived from the original on February 18, 2015. Retrieved October 8, 2008.
- ^ Jasinski, Stephen M. "Mineral Yearbook 1994: Zinc" (PDF). United States Geological Survey. Archived (PDF) from the original on October 29, 2008. Retrieved November 13, 2008.
- ^ "Diecasting Alloys". Maybrook, NY: Eastern Alloys. Archived from the original on December 25, 2008. Retrieved January 19, 2009.
- .
- ISBN 978-0-7506-5692-4. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ^ Samans, Carl Hubert (1949). Engineering Metals and Their Alloys. Macmillan Co.
- ^ ISBN 978-0-8247-9213-8. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ISBN 978-0-8317-1565-6. Archivedfrom the original on November 15, 2012. Retrieved June 26, 2012.
- ^ "Cast flywheel on old Magturbo trainer has been recalled since July 2000". Minoura. Archived from the original on March 23, 2013.
- ^ ISBN 978-0-19-514570-0.
- ISBN 978-0-306-45334-2.
- U.S. Department of Energy. Archived(PDF) from the original on February 5, 2009. Retrieved January 10, 2009.
- ^ a b c Heiserman 1992, p. 124
- (PDF) from the original on January 14, 2012.
- from the original on March 31, 2023. Retrieved September 4, 2019.
- ^ a b CRC 2006, p. 4-42
- ]
- PMID 14749112.
- ^ a b c Boudreaux, Kevin A. "Zinc + Sulfur". Angelo State University. Archived from the original on December 2, 2008. Retrieved October 8, 2008.
- ^ "Rolled and Titanium Zinc Sheet". Archived from the original on July 7, 2022. Retrieved October 21, 2022.
- ^ "Things You Should Know About Zinc Countertops". Archived from the original on October 22, 2022. Retrieved October 21, 2022.
- ^ "Guide to Zinc Countertops: Benefits of Zinc Kitchen Counters". Archived from the original on October 22, 2022. Retrieved October 21, 2022.
- ^ "Technical Information". Zinc Counters. 2008. Archived from the original on November 21, 2008. Retrieved November 29, 2008.
- ^ a b c Win, David Tin; Masum, Al (2003). "Weapons of Mass Destruction" (PDF). Assumption University Journal of Technology. 6 (4). Assumption University: 199. Archived (PDF) from the original on March 26, 2009. Retrieved April 6, 2009.
- ISBN 978-0-7876-2846-8. Archivedfrom the original on July 10, 2008. Retrieved April 6, 2009.
- ]
- ISBN 978-1-4020-4392-5.
- ^ "ZDDP Engine Oil – The Zinc Factor". Mustang Monthly. Archived from the original on September 12, 2009. Retrieved September 19, 2009.
- PMID 16721894.
- ISBN 978-0-471-26418-7.
- ISBN 978-0-470-09337-5. Archivedfrom the original on April 14, 2016.
- ISBN 978-0-19-850121-3. Archivedfrom the original on April 14, 2016.
- ISBN 978-3-13-103061-0. Archivedfrom the original on April 14, 2016.
- ^ E. Frankland, Ann. 126, 109 (1863)
- ^ E. Frankland, B. F. Duppa, Ann. 135, 25 (1865)
- .
- ^ PMID 38367035.
- ISBN 978-0-8493-1652-4.
- ^ Sanchez, Juliana (February 13, 2013). Zinc Sulphate vs. Zinc Amino Acid Chelate (ZAZO) (Report). USA Government. NCT01791608. Archived from the original on April 6, 2022. Retrieved April 6, 2022 – via U.S. National Library of Medecine.
- PMID 24826920.
- S2CID 196616666.
- PMID 11101480.
- PMID 16434472.
- PMID 15892631. Archived from the original(PDF) on August 8, 2017.
- ^ a b c d e f "Zinc – Fact Sheet for Health Professionals". Office of Dietary Supplements, US National Institutes of Health. February 11, 2016. Retrieved January 7, 2018.
- ^ PMID 22566526.
- ^ "Common Cold and Runny Nose". United States Centers for Disease Control and Prevention. September 26, 2017. Retrieved January 7, 2018.
- PMID 21846317.
- PMID 10801965.
- PMID 31613478.
- from the original on September 29, 2023. Retrieved October 16, 2023.
- S2CID 1725139.
- ^ Research, Center for Drug Evaluation and (November 16, 2021). "Questions and Answers: FDA posts deemed final order and proposed order for over-the-counter sunscreen". FDA. Archived from the original on August 16, 2022. Retrieved October 31, 2022.
- from the original on March 17, 2024, retrieved October 31, 2022
- PMID 12716335.
- ^ "Toothpastes". www.ada.org. Archived from the original on March 5, 2016. Retrieved September 27, 2020.
- S2CID 23368244.
- PMID 23919052.
- PMID 37445111.
- ^ Cotton et al. 1999, pp. 625–629
- PMID 20617034.
- from the original on May 24, 2023. Retrieved September 9, 2019.
- ^ PMID 11115789.
- ISBN 978-0-8493-5227-0. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ISBN 978-0-8493-9218-4. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- PMID 30770447.
- PMID 25468189.
- PMID 26055706.
Zn is present in up to 10% of proteins in the human proteome and computational analysis predicted that ~30% of these ~3000 Zn-containing proteins are crucial cellular enzymes, such as hydrolases, ligases, transferases, oxidoreductases, and isomerases (42,43).
- ^ S2CID 206520330.
- S2CID 22507338.
- (PDF) from the original on August 29, 2017.
- PMID 17119290.
- ^ from the original on September 19, 2017.
- ISBN 978-0-7216-4452-3.
- ^ a b Greenwood & Earnshaw 1997, pp. 1224–1225
- ISBN 978-0-8247-2449-8. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ^ a b Greenwood & Earnshaw 1997, p. 1225
- ^ Cotton et al. 1999, p. 627
- .
- PMID 29394036.
- ^ Cotton et al. 1999, p. 628
- ISBN 978-1-4288-1893-4.
- PMID 17728842.
- ^ Cotton et al. 1999, p. 629
- ISBN 978-0-07-148901-0.
- ^ PMID 2407097.
- S2CID 24589993.
- PMID 21338876.
- ^ PMID 11940571.
The human dopamine transporter (hDAT) contains an endogenous high affinity Zn2+ binding site with three coordinating residues on its extracellular face (His193, His375, and Glu396). ... Thus, when Zn2+ is co-released with glutamate, it may greatly augment the efflux of dopamine.
- PMID 30618610.
- ^ a b "Overview on Dietary Reference Values for the EU population as derived by the EFSA Panel on Dietetic Products, Nutrition and Allergies" (PDF). 2017. Archived (PDF) from the original on August 28, 2017.
- ^ Tolerable Upper Intake Levels For Vitamins And Minerals (PDF), European Food Safety Authority, 2006, archived (PDF) from the original on March 16, 2016
- ^ "Federal Register May 27, 2016 Food Labeling: Revision of the Nutrition and Supplement Facts Labels. FR page 33982" (PDF). Archived (PDF) from the original on August 8, 2016.
- ^ "Daily Value Reference of the Dietary Supplement Label Database (DSLD)". Dietary Supplement Label Database (DSLD). Archived from the original on April 7, 2020. Retrieved May 16, 2020.
- ISBN 978-0-8493-8980-1. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- ^ "Zinc content of selected foods per common measure" (PDF). USDA National Nutrient Database for Standard Reference, Release 20. United States Department of Agriculture. Archived from the original (PDF) on March 5, 2009. Retrieved December 6, 2007.
- ^ PMID 9701167.
- PMID 12949397.
- PMID 15867288.
- ^ a b "The impact of zinc supplementation on childhood mortality and severe morbidity". World Health Organization. 2007. Archived from the original on March 2, 2009.
- PMID 15705693.
- ^ Moshfegh, Alanna; Goldman, Joseph; Cleveland, Linda (2005). "NHANES 2001–2002: Usual Nutrient Intakes from Food Compared to Dietary Reference Intakes" (PDF). U.S. Department of Agriculture, Agricultural Research Service. Table A13: Zinc. Archived (PDF) from the original on July 6, 2023. Retrieved January 6, 2015.
- ^ What We Eat In America, NHANES 2013–2014 Archived February 24, 2017, at the Wayback Machine.
- PMID 12730441.
- ^ (PDF) from the original on January 14, 2017.
- S2CID 8424197.
- PMID 12612181.
- from the original on October 25, 2014.
- ^ Alloway, Brian J. (2008). "Zinc in Soils and Crop Nutrition, International Fertilizer Industry Association, and International Zinc Association". Archived from the original on February 19, 2013.
- ^ Eisler, Ronald (1993). "Zinc Hazard to Fish, Wildlife, and Invertebrates: A Synoptic Review". Contaminant Hazard Reviews (10). Laurel, Maryland: U.S. Department of the Interior, Fish and Wildlife Service. Archived (PDF) from the original on March 6, 2012.
- PMID 16472524.
- PMID 12612262.
- PMID 17222649.
- ^ Richard Martin (February 15, 2010). "Lawsuits blame denture adhesives for neurological damage (Denture adhesives cited in lawsuits)". St. Petersburg Times. Archived from the original on October 11, 2012. Retrieved December 31, 2022.
- ISBN 978-0-444-80566-9.
- ^ "FDA says Zicam nasal products harm sense of smell". Los Angeles Times. June 17, 2009. Archived from the original on June 21, 2012.
- PMID 19809895.
- PMID 10382562.
- PMID 9185931.
- PMID 3941880.
- PMID 689968.
- PMID 3767804.
Bibliography
- Chambers, William and Robert (1901). Chambers's Encyclopaedia: A Dictionary of Universal Knowledge (Revised ed.). London and Edinburgh: J. B. Lippincott Company. Archived from the original on March 17, 2024. Retrieved September 27, 2020.
- Cotton, F. Albert; Wilkinson, Geoffrey; Murillo, Carlos A.; Bochmann, Manfred (1999). Advanced Inorganic Chemistry (6th ed.). New York: John Wiley & Sons, Inc. ISBN 978-0-471-19957-1.
- David R. Lide, ed. (2006). Handbook of Chemistry and Physics (87th ed.). Boca Raton, Florida: CRC Press, Taylor & Francis Group. ISBN 978-0-8493-0487-3. Archivedfrom the original on March 17, 2024. Retrieved September 27, 2020.
- Emsley, John (2001). "Zinc". Nature's Building Blocks: An A-Z Guide to the Elements. Oxford, England, UK: Oxford University Press. pp. 499–505. ISBN 978-0-19-850340-8.
- Greenwood, N. N.; Earnshaw, A. (1997). Chemistry of the Elements (2nd ed.). Oxford: Butterworth-Heinemann. ISBN 978-0-7506-3365-9.
- Heiserman, David L. (1992). "Element 30: Zinc". Exploring Chemical Elements and their Compounds. New York: TAB Books. ISBN 978-0-8306-3018-9.
- Lehto, R. S. (1968). "Zinc". In Clifford A. Hampel (ed.). The Encyclopedia of the Chemical Elements. New York: Reinhold Book Corporation. pp. 822–830. LCCN 68-29938.
- Stwertka, Albert (1998). "Zinc". Guide to the Elements (Revised ed.). Oxford University Press. ISBN 978-0-19-508083-4.
- ISBN 978-0-7661-3872-8.
External links
- Zinc: Fact Sheet for Health Professionals from the U.S. Office of Dietary Supplements
- Zinc Statistics and Information from the U.S. Geological Survey's National Minerals Information Center
- Zinc.org - official website of the International Zinc Association, a zinc industry association
- Zinc video from the Periodic Videos series (University of Nottingham)
- ZincBind.net – a database identifying biological zinc binding sites from within the Protein Data Bank

![{\displaystyle {\ce {2ZnS + 3O2 ->[t^o] 2ZnO + 2SO2}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/686cb2d7d73dea23bc0e3431bfab4016d5fd71ed)
![{\displaystyle {\ce {ZnO+C->[950^{\circ }C]Zn+CO}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/9fdfb17e432e4fdf9bc27745fc247e68d37bbf79)
![{\displaystyle {\ce {ZnO+CO->[950^{\circ }C]Zn+CO2}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/5fddd497b421990fd58dab27d03ffeef37b088b6)

