Zopiclone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Imovane, Zimovane, Dopareel, others |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Oral tablets, 3.75 mg or 7.5mg (UK), 5 mg, 7.5 mg, or 10 mg (JP) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 75–80%[2] |
| Protein binding | 52–59% |
| Metabolism | Hepatic through CYP3A4 and CYP2E1 |
| Elimination half-life | ~5 hours (3.5–6.5 hours) ~7–9 hours for 65+ years old |
| Excretion | Urine (80%) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Zopiclone, sold under the brand name Imovane among others, is a
Zopiclone is a
In the United States, zopiclone is not commercially available,
Zopiclone is known colloquially as a "
Medical uses

Zopiclone is used for the short-term treatment of insomnia where sleep initiation or sleep maintenance are prominent symptoms. Long-term use is not recommended, as tolerance, dependence, and addiction can occur.[6][7] One low-quality study found that zopiclone is ineffective in improving sleep quality or increasing sleep time in shift workers, and more research in this area has been recommended.[8]
Cognitive behavioral therapy has been found to be superior to zopiclone in the treatment of insomnia and has been found to have lasting effects on sleep quality for at least a year after therapy.[9][10][11][12]
Specific populations
Elderly
Zopiclone, similar to other benzodiazepines and nonbenzodiazepine hypnotic drugs, causes impairments in body balance and standing steadiness in individuals who wake up at night or the next morning. Falls and hip fractures are frequently reported. The combination with alcohol consumption increases these impairments. Partial, but incomplete tolerance develops to these impairments.[13] Zopiclone increases
An extensive review of the medical literature regarding the management of insomnia and the elderly found that considerable evidence of the effectiveness and lasting benefits of nondrug treatments for insomnia exist. Compared with the benzodiazepines, the nonbenzodiazepine sedative-hypnotics, such as zopiclone, offer few if any advantages in efficacy or tolerability in elderly persons. Newer agents such as the melatonin receptor agonists may be more suitable and effective for the management of chronic insomnia in elderly people. Long-term use of sedative-hypnotics for insomnia lacks an evidence base and is discouraged for reasons that include concerns about such potential adverse drug effects as cognitive impairment (anterograde amnesia), daytime sedation, motor incoordination, and increased risk of motor vehicle accidents and falls. In addition, the effectiveness and safety of long-term use of nonbenzodiazepine hypnotic drugs remains to be determined.[17]
Liver disease
Patients with liver disease eliminate zopiclone much more slowly than normal patients and in addition experience exaggerated pharmacological effects of the drug.[18]
Adverse reactions
Sleeping pills, including zopiclone, have been associated with an increased risk of death.[19] The British National Formulary states adverse reactions as follows: "taste disturbance (some report a metallic taste); less commonly nausea, vomiting, dizziness, drowsiness, dry mouth, headache; rarely amnesia, confusion, depression, hallucinations, nightmares; very rarely light headedness, incoordination, paradoxical effects [...] and sleep-walking also reported".[20]
Contraindications
Zopiclone causes impaired driving skills similar to those of benzodiazepines. Long-term users of hypnotic drugs for sleep disorders develop only partial tolerance to adverse effects on driving, with users of hypnotic drugs even after one year of use still showing an increased motor vehicle accident rate.[21] Patients who drive motor vehicles should not take zopiclone as there is a significantly increased risk of accidents in zopiclone users.[22] Zopiclone induces impairment of psychomotor function.[23][24] Driving or operating machinery should be avoided after taking zopiclone as effects can carry over to the next day, including impaired hand-eye coordination.[25][26]
A
EEG and sleep
It causes similar alterations on EEG readings and
Overdose
Zopiclone is sometimes used as a method of suicide.
Death certificates show the number of zopiclone-related deaths is on the rise.
Interactions
Zopiclone also interacts with trimipramine and caffeine.[49][50]
Alcohol has an additive effect when combined with zopiclone, enhancing the adverse effects including the overdose potential of zopiclone significantly.[51][52] Due to these risks and the increased risk for dependence, alcohol should be avoided when using zopiclone.[51]
Pharmacology
The therapeutic pharmacological properties of zopiclone include
APharmacokinetics
After oral administration, zopiclone is rapidly absorbed, with a bioavailability around 75–80%. Time to peak plasma concentration is 1–2 hours. A high-fat meal preceding zopiclone administration does not change absorption (as measured by AUC), but reduces peak plasma levels and delays its occurrence, thus may delay the onset of therapeutic effects.
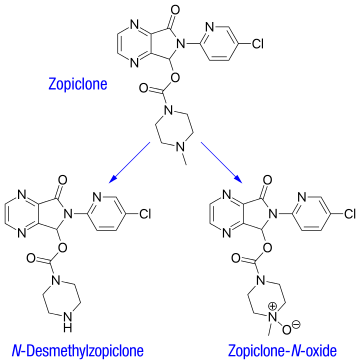
The plasma protein-binding of zopiclone has been reported to be weak, between 45 and 80% (mean 52–59%). It is rapidly and widely distributed to body tissues, including the brain, and is excreted in urine, saliva, and breast milk. Zopiclone is partly extensively metabolized in the liver to form an active N-demethylated derivative (N-desmethylzopiclone) and an inactive zopiclone-N-oxide. Hepatic enzymes playing the most significant role in zopiclone metabolism are CYP3A4 and CYP2E1. In addition, about 50% of the administered dose is decarboxylated and excreted via the lungs. In urine, the N-demethyl and N-oxide metabolites account for 30% of the initial dose. Between 7 and 10% of zopiclone is recovered from the urine, indicating extensive metabolism of the drug before excretion. The terminal elimination half-life of zopiclone ranges from 3.5 to 6.5 hours (5 hours on average).[2]
The
The pharmacokinetics of zopiclone are altered by aging and are influenced by renal and hepatic functions.[69] In severe chronic kidney failure, the area under the curve value for zopiclone was larger and the half-life associated with the elimination rate constant longer, but these changes were not considered to be clinically significant.[70] Sex and race have not been found to interact with pharmacokinetics of zopiclone.[2]
Chemistry
The melting point of zopiclone is 178 °C.[71] Zopiclone's solubility in water, at room temperature (25 °C) are 0.151 mg/mL.[71] The logP value of zopiclone is 0.8.[71]
Detection in biological fluids
Zopiclone may be measured in blood, plasma, or urine by chromatographic methods. Plasma concentrations are typically less than 100 μg/L during therapeutic use, but frequently exceed 100 μg/L in automotive vehicle operators arrested for impaired driving ability and may exceed 1000 μg/L in acutely poisoned patients. Post mortem blood concentrations are usually in a range of 400 to 3900 μg/L in victims of fatal acute overdose.[72][73][74]
History
Zopiclone was developed and first introduced in 1986 by
Zopiclone, as traditionally sold worldwide, is a
Society and culture
Recreational use
Zopiclone has the potential for non-medical use, dosage escalation, and drug dependence. It is taken orally and sometimes intravenously when used non-medically, and often combined with alcohol to achieve euphoria. Patients abusing the drug are also at risk of dependence. Withdrawal symptoms can be seen after long-term use of normal doses even after a gradual reduction regimen. The Compendium of Pharmaceuticals and Specialties recommends zopiclone prescriptions not exceed 7 to 10 days, owing to concerns of addiction, tolerance, and physical dependence.
Zopiclone and other sedative hypnotic drugs are detected frequently in cases of people suspected of driving under the influence of drugs. Other sedating drugs, including benzodiazepines and zolpidem, are also found in high numbers of suspected drugged drivers. Many drivers have blood levels far exceeding the therapeutic dose range and often in combination with alcohol, illegal, or addictive
Zopiclone has crosstolerance with barbiturates and is able to suppress barbiturate withdrawal symptoms. It is frequently self-administered intravenously in studies on monkeys, suggesting a high risk of addictive potential.[89]
Zopiclone is in the top ten medications obtained using a false prescription in France.[2]
References
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ a b c d "Assessment of Zopiclone" (PDF). World Health Organization. Essential Medicines and Health Products. World Health Organization. 2006. p. 9 (Section 5. Pharmacokinetics). Retrieved 5 December 2015.
- ^ "Zopiclone consumer information from". Drugs.com. Retrieved 2013-06-06.
- ^ "Chapter 3 - The technologies, section 3.4", Clinical need and practice - Guidance on the use of zaleplon, zolpidem and zopiclone for the short-term management of insomnia, National Institute for Health and Care Excellence (NICE), 28 April 2004, Technology appraisal guidance [TA77],
This guidance will be reviewed if there is new evidence.
Current as of 8 June 2023 - PMID 2744064.
- S2CID 40188442.
- PMID 17652991.
- PMID 25113164.
- PMID 17089469.
- PMID 14609970.
- PMID 16804151.
- PMID 14960254.
- PMID 20171127.
- PMID 8127419.
- S2CID 13440208.
- PMID 16916422.
- PMID 16860264.
- PMID 6626417.
- S2CID 7946506.
- ^ "Zopiclone", British National Formulary, National Institute for Health and Care Excellence, 19 September 2016, archived from the original on 9 October 2016, retrieved 2 October 2016
- S2CID 26351598.
- S2CID 40825194.
- PMID 16045197.
- S2CID 36841164.
- PMID 7188379.
- PMID 3326113.
- PMID 14692469.
- PMID 2306331.
- S2CID 25259646.
- PMID 3505734.
- PMID 8290860.
- S2CID 54285586.
- PMID 15037809.
- PMID 8939015.
- ^ Buckley NA, Dawson AH, Whyte IM, McManus P, Ferguson N.Correlations between prescriptions and drugs taken in self-poisoning: Implications for prescribers and drug regulation.Med J Aust (in press)
- PMID 7866122.
- PMID 9068198.
- PMID 8837952.
- PMID 9893443.
- PMID 8868406.
- S2CID 41701825.
- PMID 8039775.
- PMID 2353332.
- PMID 2814922.
- ISBN 978-0-7817-2845-4.
- S2CID 24102880.
- PMID 9183949.
- PMID 11569575.
- PMID 6743780.
- PMID 1351673.
- ^ PMID 2201724.
- S2CID 37777007.
- PMID 7833227.
- S2CID 20916689.
- PMID 9159561.
- PMID 10460808. Archived from the originalon 2005-04-17. Retrieved 2008-12-16.
- S2CID 38894701.[permanent dead link]
- S2CID 40503805.
- S2CID 46510829.
- PMID 12871032.
- PMID 2581904.
- PMID 2860074.
- S2CID 10888200.
- S2CID 35732735.
- PMID 6135616.
- PMID 2905900.
- PMID 6142468.
- PMID 6322210.
- PMID 7188377.
- PMID 2387982.
- ^ a b c "Zopiclone". pubchem.ncbi.nlm.nih.gov. U.S. National Library of Medicine. Retrieved 11 June 2018.
- PMID 15329838.
- PMID 19393793.
- ^ Baselt R (2008). Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Foster City, CA: Biomedical Publications. pp. 1677–1679.
- PMID 10674059.
- PMID 8398600.
- PMID 7905394.
- PMID 18077750.
- PMID 16336040.
- S2CID 31103719.
- PMID 9656789.
- S2CID 42630879.
- PMID 9657802.
- S2CID 33400840. Archived from the original(PDF) on 2016-03-03. Retrieved 2008-12-18.
- S2CID 25511804.
- PMID 10494203.
- S2CID 45013069.
- S2CID 46445345.
- PMID 6892368.
External links
- Detailed pharmacological information Archived 2006-04-04 at the Wayback Machine
- Scheduling recommendation (PDFfile)
- Details on scheduling
- Erowid zopiclone vault
- Support for zopiclone dependency/addiction Archived 2015-04-02 at the Wayback Machine
