Zwitterion
In chemistry, a zwitterion (/ˈtsvɪtəˌraɪən/ TSVIT-ə-rye-ən; from German Zwitter [ˈtsvɪtɐ] 'hermaphrodite'), also called an inner salt or dipolar ion,[1] is a molecule that contains an equal number of positively and negatively charged functional groups.[2] With amino acids, for example, in solution a chemical equilibrium will be established between the "parent" molecule and the zwitterion.
Amino acids

Tautomerism of amino acids follows this stoichiometry:
- RCH(NH2)CO2H ⇌ RCH(N+H3)CO−2
The ratio of the concentrations of the two species in solution is independent of pH.
It has been suggested, on the basis of theoretical analysis, that the zwitterion is stabilized in aqueous solution by
The pKa values for deprotonation of the common amino acids span the approximate range 2.15±0.2. This is also consistent with the zwitterion being the predominant isomer that is present in an aqueous solution. For comparison, the simple carboxylic acid propionic acid (CH3CH2CO2H) has a pKa value of 4.88.
Other compounds
-
Sulfamic acid isomers, with the zwitterion (right)
-
Structure ofH4EDTA
Sulfamic acid crystallizes in the zwitterion form.[6]
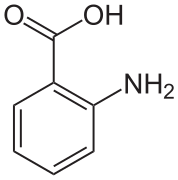
In crystals of anthranilic acid there are two molecules in the unit cell. One molecule is in the zwitterion form, the other is not.[7]
In the solid state,
In psilocybin, the proton on the dimethyl amino group is labile and may jump to the phosphate group to form a compound which is not a zwitterion.
Theoretical studies

Insight to the equilibrium in solution may be gained from the results of theoretical calculations. For example, pyridoxal phosphate, a form of vitamin B6, in aqueous solution is predicted to have an equilibrium favoring a tautomeric form in which a proton is transferred from the phenolic -OH group to the nitrogen atom.[9]
Because tautomers are different compounds, they sometimes have different enough structures that they can be detected independently in their mixture. This allows experimental analysis of the equilibrium.[10]
Betaines and similar compounds
The compound
Other examples of permanent zwitterions include phosphatidylcholines, which also contain a quaternary nitrogen atom, but with a negatively-charged phosphate group in place of a carboxylate group; sulfobetaines, which contain a quaternary nitrogen atom and a negatively charged sulfonate group;[12] and pulmonary surfactants such as dipalmitoylphosphatidylcholine. Lauramidopropyl betaine is the major component of cocamidopropyl betaine.
-
Trimethylglycine (trivial name betaine)
-
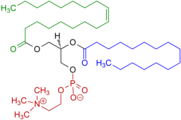
Example of a phosphatidylcholine
Conjugated zwitterions
Strongly polarized conjugated compounds (conjugated zwitterions) are typically very reactive, share diradical character, activate strong bonds and small molecules, and serve as transient intermediates in catalysis.[13] Donor-acceptor entities are of vast use in photochemistry (photoinduced electron transfer), organic electronics, switching and sensing.
See also
References
- ^ "Zwitterion". Chemistry LibreTexts. 2015-11-03. Retrieved 2022-02-11.
- ISBN 0-03-035523-0.
- Fundamentals of Analytical Chemistry (9th ed.). 2013. pp. 415–416. ISBN 978-1-285-60719-1.
- Fundamentals of Analytical Chemistry (9th ed.). 2013. pp. 415–416.
- .
- .
- PMID 16479267.
- .
- .
- .
- PMID 12662064.
- .
- ISBN 1-57259-153-6.
- PMID 677454.
- S2CID 235220781.