Huntington's disease
| Huntington's disease | |
|---|---|
| Other names | Huntington's chorea |
Supportive care[2] | |
| Medication | Tetrabenazine[3] |
| Prognosis | 15–20 years from onset of symptoms[4] |
| Frequency | 4–15 in 100,000 (European descent)[1] |
| Named after | George Huntington |
Huntington's disease (HD), also known as Huntington's chorea, is an incurable
HD is typically
No cure for HD is known, and full-time care is required in the later stages.
The earliest known description of the disease was in 1841 by American physician Charles Oscar Waters.[15] The condition was described in further detail in 1872 by American physician George Huntington.[15] The genetic basis was discovered in 1993 by an international collaborative effort led by the Hereditary Disease Foundation.[16][17] Research and support organizations began forming in the late 1960s to increase public awareness, provide support for individuals and their families and promote research.[17][18] Research directions include determining the exact mechanism of the disease, improving animal models to aid with research, testing of medications and their delivery to treat symptoms or slow the progression of the disease, and studying procedures such as stem-cell therapy with the goal of replacing damaged or lost neurons.[16]
Signs and symptoms
The most characteristic initial physical symptoms are jerky, random, and uncontrollable movements called chorea.[10] Many people are not aware of their involuntary movements, or impeded by them.[1] Chorea may be initially exhibited as general restlessness, small unintentionally initiated or uncompleted motions, lack of coordination, or slowed saccadic eye movements.[26] These minor motor abnormalities usually precede more obvious signs of motor dysfunction by at least three years.[27] The clear appearance of symptoms such as rigidity, writhing motions, or abnormal posturing appear as the disorder progresses.[26] These are signs that the system in the brain that is responsible for movement has been affected.[28] Psychomotor functions become increasingly impaired, such that any action that requires muscle control is affected. When muscle control is affected such as rigidity or muscle contracture this is known as dystonia. Dystonia is a neurological hyperkinetic movement disorder that results in twisting or repetitive movements, that may resemble a tremor. Common consequences are physical instability, abnormal facial expression, and difficulties chewing, swallowing, and speaking.[26] Sleep disturbances and weight loss are also associated symptoms.[29] Eating difficulties commonly cause weight loss and may lead to malnutrition.[30][31] Weight loss is common in people with Huntington's disease, and it progresses with the disease. Juvenile HD generally progresses at a faster rate with greater cognitive decline, and chorea is exhibited briefly, if at all; the Westphal variant of slowness of movement, rigidity, and tremors is more typical in juvenile HD, as are seizures.[26][29]
Cognitive abilities are progressively impaired and tend to generally decline into
Reported
Mutant huntingtin is expressed throughout the body and associated with abnormalities in peripheral tissues that are directly caused by such expression outside the brain. These abnormalities include
Genetics
Everyone has two copies of the
Genetic mutation
HD is one of several
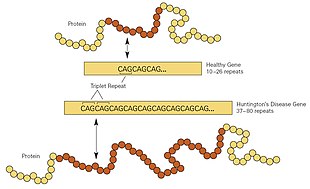
| Repeat count | Classification | Disease status | Risk to offspring |
|---|---|---|---|
| <27 | Normal | Will not be affected | None |
| 27–35 | Intermediate | Will not be affected | Elevated, but <50% |
| 36–39 | Reduced Penetrance | May or may not be affected | 50% |
| 40+ | Full penetrance | Will be affected | 50% |
Generally, people have fewer than 36 repeated glutamines in the polyQ region, which results in the production of the
Inheritance
Huntington's disease has
Trinucleotide CAG repeats numbering over 28 are unstable during replication, and this instability increases with the number of repeats present.[26] This usually leads to new expansions as generations pass (dynamic mutations) instead of reproducing an exact copy of the trinucleotide repeat.[26] This causes the number of repeats to change in successive generations, such that an unaffected parent with an "intermediate" number of repeats (28–35), or "reduced penetrance" (36–40), may pass on a copy of the gene with an increase in the number of repeats that produces fully penetrant HD.[26] The earlier age of onset and greater severity of disease in successive generations due to increases in the number of repeats is known as genetic anticipation.[1] Instability is greater in spermatogenesis than oogenesis;[26] maternally inherited alleles are usually of a similar repeat length, whereas paternally inherited ones have a higher chance of increasing in length.[26][40] Rarely is Huntington's disease caused by a new mutation, where neither parent has over 36 CAG repeats.[41]
In the rare situations where both parents have an expanded HD gene, the risk increases to 75%, and when either parent has two expanded copies, the risk is 100% (all children will be affected). Individuals with
Mechanisms
Huntingtin protein interacts with over 100 other proteins, and appears to have multiple functions.
Huntingtin function
Htt is
Cellular changes
The toxic action of mHtt may manifest and produce the HD pathology through multiple cellular changes.
Several pathways by which mHtt may cause cell death have been identified. These include effects on chaperone proteins, which help fold proteins and remove misfolded ones; interactions with caspases, which play a role in the process of removing cells; the toxic effects of glutamine on nerve cells; impairment of energy production within cells; and effects on the expression of genes.[13][51]
Mutant huntingtin protein has been found to play a key role in
Glutamine is known to be
Macroscopic changes
Initially, damage to the brain is regionally specific with the
The basal ganglia play a key role in movement and behavior control. Their functions are not fully understood, but theories propose that they are part of the cognitive
Transcriptional dysregulation
CREB-binding protein (CBP), a transcriptional coregulator, is essential for cell function because as a coactivator at a significant number of promoters, it activates the transcription of genes for survival pathways.[51] CBP contains an acetyltransferase domain to which HTT binds through its polyglutamine-containing domain.[59] Autopsied brains of those who had Huntington's disease also have been found to have incredibly reduced amounts of CBP.[60] In addition, when CBP is overexpressed, polyglutamine-induced death is diminished, further demonstrating that CBP plays an important role in Huntington's disease and neurons in general.[51]
Diagnosis
Diagnosis of the onset of HD can be made following the appearance of physical symptoms specific to the disease.[26] Genetic testing can be used to confirm a physical diagnosis if no family history of HD exists. Even before the onset of symptoms, genetic testing can confirm if an individual or embryo carries an expanded copy of the trinucleotide repeat (CAG) in the HTT gene that causes the disease. Genetic counseling is available to provide advice and guidance throughout the testing procedure and on the implications of a confirmed diagnosis. These implications include the impact on an individual's psychology, career, family-planning decisions, relatives, and relationships. Despite the availability of pre-symptomatic testing, only 5% of those at risk of inheriting HD choose to do so.[26]
Clinical
A
Predictive genetic testing
Because HD follows an autosomal dominant pattern of inheritance, a strong motivation exists for individuals who are at risk of inheriting it to seek a diagnosis. The genetic test for HD consists of a blood test, which counts the numbers of CAG repeats in each of the HTT alleles.
- At 40 or more CAG repeats, full positive test" or "positive result" generally refers to this case. A positive result is not considered a diagnosis, since it may be obtained decades before the symptoms begin. However, a negative test means that the individual does not carry the expanded copy of the gene and will not develop HD.[26] The test will tell a person who originally had a 50% chance of inheriting the disease if their risk goes up to 100% or is eliminated. Persons who test positive for the disease will develop HD sometime within their lifetimes, provided they live long enough for the disease to appear.[26]
- At 36 to 39 repeats, incomplete or reduced penetrance allele (RPA) may cause symptoms, usually later in the adult life.[65] The maximum risk is 60% that a person with an RPA will be symptomatic at age 65, and 70% at 75.[65]
- At 27 to 35 repeats, intermediate allele (IA), or large normal allele, is not associated with symptomatic disease in the tested individual, but may expand upon further inheritance to give symptoms in offspring.[65]
- With 26 or fewer repeats, the result is not associated with HD.[65]
Testing before the onset of symptoms is a life-changing event and a very personal decision.[26] The main reason given for choosing to test for HD is to aid in career and family decisions.[26] Predictive testing for Huntington's disease has been available via linkage analysis (which requires testing multiple family members) since 1986 and via direct mutation analysis since 1993.[66] At that time, surveys indicated that 50–70% of at-risk individuals would have been interested in receiving testing, but since predictive testing has been offered far fewer choose to be tested.[67] Over 95% of individuals at risk of inheriting HD do not proceed with testing, mostly because it has no treatment.[26] A key issue is the anxiety an individual experiences about not knowing whether they will eventually develop HD, compared to the impact of a positive result.[26] Irrespective of the result, stress levels are lower two years after being tested, but the risk of suicide is increased after a positive test result.[26] Individuals found to have not inherited the disorder may experience survivor guilt about family members who are affected.[26] Other factors taken into account when considering testing include the possibility of discrimination and the implications of a positive result, which usually means a parent has an affected gene and that the individual's siblings will be at risk of inheriting it.[26] In one study, genetic discrimination was found in 46% of individuals at risk for Huntington's disease. It occurred at higher rates within personal relationships than health insurance or employment relations.[68] Genetic counseling in HD can provide information, advice and support for initial decision-making, and then, if chosen, throughout all stages of the testing process.[69] Because of the implications of this test, patients who wish to undergo testing must complete three counseling sessions which provide information about Huntington's.[70]
Counseling and guidelines on the use of genetic testing for HD have become models for other genetic disorders, such as autosomal dominant
Preimplantation genetic diagnosis
Embryos produced using in vitro fertilization may be genetically tested for HD using preimplantation genetic diagnosis. This technique, where one or two cells are extracted from a typically 4- to 8-cell embryo and then tested for the genetic abnormality, can then be used to ensure embryos affected with HD genes are not implanted, so any offspring will not inherit the disease. Some forms of preimplantation genetic diagnosis—non-disclosure or exclusion testing—allow at-risk people to have HD-free offspring without revealing their own parental genotype, giving no information about whether they themselves are destined to develop HD. In exclusion testing, the embryo's DNA is compared with that of the parents and grandparents to avoid inheritance of the chromosomal region containing the HD gene from the affected grandparent. In nondisclosure testing, only disease-free embryos are replaced in the uterus while the parental genotype and hence parental risk for HD are never disclosed.[74][75]
Prenatal testing
Obtaining a
In addition, in at-risk pregnancies due to an affected male partner, noninvasive prenatal diagnosis can be performed by analyzing cell-free fetal DNA in a blood sample taken from the mother (via venipuncture) between six and 12 weeks of pregnancy.[65] It has no procedure-related risk of miscarriage.[65]
Differential diagnosis
About 99% of HD diagnoses based on the typical symptoms and a
Management

Treatments are available to reduce the severity of some HD symptoms.
Therapy
Weight loss and problems in eating due to
People with Huntington's disease may see a
Additionally, an increasing number of people with HD are turning to palliative care, which aims to improve quality of life through the treatment of the symptoms and stress of serious illness, in addition to their other treatments.[86]
Medications
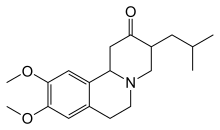
Other drugs that help to reduce chorea include
Psychiatric symptoms can be treated with medications similar to those used in the general population.[26][81] Selective serotonin reuptake inhibitors and mirtazapine have been recommended for depression, while atypical antipsychotics are recommended for psychosis and behavioral problems.[81] Specialist neuropsychiatric input is recommended since people may require long-term treatment with multiple medications in combination.[26]
Plant-based medications
There has been a number of alternative therapies experimented in ayurvedic medicine with plant-based products, although none have provided good evidence of efficacy. A recent study showed that the stromal processing peptidase (SPP), a synthetic enzyme found in plant chloroplasts, prevented the aggregation of proteins associated with Huntington's disease.[91] However, repeat studies and clinical validation are needed to confirm its true therapeutic potential.
Education
The families of individuals, and society at large, who have inherited or are at risk of inheriting HD have generations of experience of HD but may be unaware of recent breakthroughs in understanding the disease, and of the availability of genetic testing. Genetic counseling benefits these individuals by updating their knowledge, seeking to dispel any unfounded beliefs that they may have, and helping them consider their future options and plans. The Patient Education Program for Huntington's Disease has been created to help educate family members, caretakers, and those diagnosed with Huntington's disease.[92] Also covered is information concerning family planning choices, care management, and other considerations.[26][93]
Prognosis
The length of the trinucleotide repeat accounts for 60% of the variation of the age of symptoms onset and their rate of progress. A longer repeat results in an earlier age of onset and a faster progression of symptoms.[26][94] Individuals with more than sixty repeats often develop the disease before age 20, while those with fewer than 40 repeats may remain asymptomatic.[95] The remaining variation is due to environmental factors and other genes that influence the mechanism of the disease.[26]
Life expectancy in HD is generally around 10 to 30 years following the onset of visible symptoms.
Epidemiology
The late onset of Huntington's disease means it does not usually affect reproduction.
Until the discovery of a genetic test, statistics could only include
History

In centuries past, various kinds of chorea were at times called by names such as Saint Vitus' dance, with little or no understanding of their cause or type in each case.
The first definite mention of HD was in a letter by
The first thorough description of the disease was by George Huntington in 1872. Examining the combined medical history of several generations of a family exhibiting similar symptoms, he realized their conditions must be linked; he presented his detailed and accurate definition of the disease as his first paper. Huntington described the exact pattern of inheritance of autosomal dominant disease years before the rediscovery by scientists of Mendelian inheritance.
Of its hereditary nature. When either or both the parents have shown manifestations of the disease ... one or more of the offspring almost invariably suffer from the disease ... But if by any chance these children go through life without it, the thread is broken and the grandchildren and great-grandchildren of the original shakers may rest assured that they are free from the disease.[109][115]
Sir William Osler was interested in the disorder and chorea in general, and was impressed with Huntington's paper, stating, "In the history of medicine, there are few instances in which a disease has been more accurately, more graphically or more briefly described."[116][111][117] Osler's continued interest in HD, combined with his influence in the field of medicine, helped to rapidly spread awareness and knowledge of the disorder throughout the medical community.[111] Great interest was shown by scientists in Europe, including Louis Théophile Joseph Landouzy, Désiré-Magloire Bourneville, Camillo Golgi, and Joseph Jules Dejerine, and until the end of the century, much of the research into HD was European in origin.[111] By the end of the 19th century, research and reports on HD had been published in many countries and the disease was recognized as a worldwide condition.[111]
During the rediscovery of Mendelian inheritance at the turn of the 20th century, HD was used tentatively as an example of autosomal dominant inheritance.
The search for the cause of this condition was enhanced considerably in 1968, when the
The foundation was involved in the recruitment of more than 100 scientists in the US-Venezuela Huntington's Disease Collaborative Project, which over a 10-year period from 1979, worked to locate the genetic cause.
In the same time, key discoveries concerning the mechanisms of the disorder were being made, including the findings by Anita Harding's research group on the effects of the gene's length.[128]
Modelling the disease in various types of animals, such as the
The networks of care and support that had developed in Venezuela and Colombia during the research projects there in the 1970s through 2000s were eventually eroded by various forces, such as the ongoing crisis in Venezuela and the death of a lead researcher in Colombia (Jorge Daza Barriga).[130] Doctors are working toward rekindling these networks because the people who have contributed to the science of Huntington's disease by participating in these studies deserve adequate follow-up care; societies elsewhere in the world who benefit from the scientific advances thus achieved owe at least that much to those who participated in the research.[130]
The condition was formerly called Huntington's chorea, but this term has been replaced by Huntington's disease because not all patients develop chorea and due to the importance of cognitive and behavioral problems.[131]
Society and culture
Ethics
The development of an accurate diagnostic test for Huntington's disease has caused social, legal, and ethical concerns over access to and use of a person's results.[135][136] Many guidelines and testing procedures have strict procedures for disclosure and confidentiality to allow individuals to decide when and how to receive their results and also to whom the results are made available.[26] Insurance companies and businesses are faced with the question of whether to use genetic test results when assessing an individual, such as for life insurance or employment. The United Kingdom's insurance companies agreed with the Department of Health and Social Care that until 2017 customers would not need to disclose predictive genetics tests to them, but this agreement explicitly excluded the government-approved test for Huntington's when writing policies with a value over £500,000.[137][138] As with other untreatable genetic conditions with a later onset, it is ethically questionable to perform presymptomatic testing on a child or adolescent since there would be no medical benefit for that individual. There is consensus for testing only individuals who are considered cognitively mature, although there is a counter-argument that parents have a right to make the decision on their child's behalf. With the lack of effective treatment, testing a person under legal age who is not judged to be competent is considered unethical in most cases.[49][139][140]
There are ethical concerns related to
Support organizations

In 1968, after experiencing HD in his wife's family, Dr. Milton Wexler was inspired to start the Hereditary Disease Foundation (HDF), with the aim of curing genetic illnesses by coordinating and supporting research.[17] The foundation and Wexler's daughter, Nancy Wexler, were key parts of the research team in Venezuela which discovered the HD gene.[17]
At roughly the same time as the HDF formed, Marjorie Guthrie helped to found the committee to Combat Huntington's Disease (now the Huntington's Disease Society of America), after her husband, folk singer-songwriter Woody Guthrie died from complications of HD.[18]
Since then, support and research organizations have formed in many countries around the world and have helped to increase public awareness of HD. A number of these collaborate in umbrella organizations, like the International Huntington Association and the European HD network.[142] Many support organizations hold an annual HD awareness event, some of which have been endorsed by their respective governments. For example, 6 June is designated "National Huntington's Disease Awareness Day" by the US Senate.[143] Many organizations exist to support and inform those affected by HD, including the Huntington's Disease Association in the UK. The largest funder of research is provided by the Cure Huntington's Disease Initiative Foundation (CHDI).[144]
Research directions
Research into the mechanism of HD is focused on identifying the functioning of Htt, how mHtt differs or interferes with it, and the brain pathology that the disease produces.
Research is being conducted using many approaches to either prevent Huntington's disease or slow its progression.
The CHDI Foundation funds a great many research initiatives providing many publications.[148] The CHDI foundation is the largest funder of Huntington's disease research globally and aims to find and develop drugs that will slow the progression of HD.[144][149] CHDI was formerly known as the High Q Foundation. In 2006, it spent $50 million on Huntington's disease research.[144] CHDI collaborates with many academic and commercial laboratories globally and engages in oversight and management of research projects as well as funding.[150]
Reducing huntingtin production
Increasing huntingtin clearance
Another strategy to reduce the level of mutant huntingtin is to increase the rate at which cells are able to clear it.[162] As mHtt (and many other protein aggregates) are degraded by autophagy, increasing the rate of autophagy has the potential to reduce levels of mHtt and thereby ameliorate disease.[163] Pharmacological and genetic inducers of autophagy have been tested in a variety of Huntington's disease models; many have been shown to reduce mHtt levels and decrease toxicity.[162]
Improving cell survival
Among the approaches aimed at improving cell survival in the presence of mutant huntingtin are correction of
Neuronal replacement
Stem-cell therapy is used to replace damaged neurons by transplantation of stem cells into affected regions of the brain. Experiments in animal models (rats and mice only) have yielded positive results.[164]
Whatever their future therapeutic potential, stem cells are already a valuable tool for studying Huntington's disease in the laboratory.[165]
Ferroptosis
Clinical trials
In 2020, there were 197
See also
References
- ^ PMID 25432725.
- ^ PMID 20301482.
- ^ PMID 24366610.
- ^ a b c d e f g h "Huntington's Disease Information Page". National Institute of Neurological Disorders and Stroke. Archived from the original on 13 December 2020. Retrieved 14 December 2020.
- ^ PMID 22902173.
- ISBN 978-0-323-07699-9.
- ^ "Huntington's disease - Treatment and support". National Health Service UK. 23 October 2017. Archived from the original on 6 May 2023. Retrieved 6 May 2023.
- ^ from the original on 13 November 2020. Retrieved 8 November 2020 – via protein.bio.msu.ru.
- ^ PMID 30542906.
- ^ ISBN 978-0-323-35317-5.
- ISBN 978-0-87893-695-3.
- ^ S2CID 8272667.
- ^ S2CID 25759303.
- ^ "Aspiration Pneumonia: What It Is, Causes, Diagnosis, Treatment". Cleveland Clinic. Archived from the original on 12 June 2023. Retrieved 12 June 2023.
- ^ PMID 26056609.
- ^ a b "About Huntington's Disease". Genome.gov. Archived from the original on 9 January 2021. Retrieved 13 January 2021.
- ^ a b c d "History of the HDF". Hereditary Disease Foundation. Archived from the original on 19 November 2015. Retrieved 18 November 2015.
- ^ a b "History and Genetics of Huntington's Disease | Huntington's Disease Society of America". March 2019. Archived from the original on 1 December 2020. Retrieved 14 December 2020.
- ^ PMID 29587954.
- ^ a b "Huntington's disease - Symptoms and causes". Mayo Clinic. Archived from the original on 5 March 2018. Retrieved 13 December 2022.
- ^ "Huntington's disease". www.nhsinform.scot. Archived from the original on 12 July 2020. Retrieved 12 July 2020.
- ^ from the original on 10 February 2009. Retrieved 22 November 2020.
- ISBN 978-0-89042-554-1.
- ISBN 978-0-19-851060-4.
- ^ Wagle AC, Wagle SA, Marková IS, Berrios GE (2000). "Psychiatric Morbidity in Huntington's disease". Neurology, Psychiatry and Brain Research (8): 5–16.
- ^ S2CID 46151626.
- ISBN 978-0-19-851060-4.
- ^ PMID 16496032. Archived from the original(PDF) on 23 March 2016. Retrieved 17 September 2008.
- ^ PMID 29218782.
- S2CID 26109381.
- ^ "Booklet by the Huntington Society of Canada" (PDF). Caregiver's Handbook for Advanced-Stage Huntington Disease. HD Society of Canada. 11 April 2007. Archived from the original (PDF) on 25 June 2008. Retrieved 10 August 2008.
- ^ PMID 18070848.
- ISBN 978-1-4377-0434-1.
- S2CID 14419437.
- PMID 18473821.
- S2CID 20523093.
- PMID 11553930.
- ISBN 978-0-86577-958-7.
- ^ "Sex Linked". Genome.gov. Archived from the original on 14 April 2022. Retrieved 13 December 2022.
- PMID 2972838.
- S2CID 26007984.
- S2CID 4312171.
- PMID 12615650.
- PMID 15383276.
- PMID 25813218.
- ^ PMID 28785371.
- ^ S2CID 10119487.
- ^ S2CID 28435830.
- ^ PMID 2136787.
- S2CID 189944779.
- ^ S2CID 12474262.
- PMID 27662334.
- PMID 27069383.
- PMID 28817209.
- ISBN 978-0-87893-742-4. Archivedfrom the original on 18 February 2009. Retrieved 1 April 2009.
- PMID 17965655.
- ^ PMID 10923984.
- ^ Duffy J (2013). Motor Speech Disorders: Substrates, Differential Diagnosis, and Management (3rd ed.). St. Louis, Missouri: Elsevier. pp. 196–7.
- from the original on 1 August 2020. Retrieved 28 June 2019.
- PMID 9478975.
- ^ Gaillard F (1 May 2007). "Huntington's disease". Radiology picture of the day. www.radpod.org. Archived from the original on 22 October 2007. Retrieved 24 July 2009.
- PMID 19111470.
- ^ "Unified Huntington's Disease Rating Scale (UHDRS)". UHDRS and Database. HSG. 1 February 2009. Archived from the original on 11 August 2015. Retrieved 14 April 2009.
- PMID 15717026.
- ^ PMID 23377865.
- PMID 27165004.
- S2CID 15447709.
- PMID 20468061.
- PMID 11575847.
- S2CID 40182214.
- ^ S2CID 39581496.
- S2CID 28018134.
- PMID 22990145.
- S2CID 45703511.
- S2CID 33967835.
- ^ "Predictive Testing for Huntington's Disease". 2011. Archived from the original on 22 January 2013. Retrieved 7 May 2013.
- S2CID 9382420.
- ^ "Guidelines for Genetic Testing for Huntington's Disease". Heredity Disease Foundation. Archived from the original on 26 June 2015. Retrieved 7 May 2013.
- ^ S2CID 9052603.
- S2CID 42386743. Archived from the originalon 8 October 2011.
- ^ S2CID 1956458.
- PMID 18390785.
- ^ a b "EHDN Physiotherapy Guidance Document" (PDF). European HD Network Physiotherapy Working Group. Archived from the original (PDF) on 4 March 2016. Retrieved 15 November 2015.
- from the original on 9 August 2020. Retrieved 10 May 2012.
- PMID 21960468.
- PMID 17505405.
- ^ "FDA Approves First Drug for Treatment of Chorea in Huntington's Disease". U.S. Food and Drug Administration. 15 August 2008. Archived from the original on 21 August 2008. Retrieved 10 August 2008.
- PMID 27988871.
- S2CID 84183892.
- ^ Center for Drug Evaluation Research (17 July 2019). "In Pursuit of Tardive Dyskinesia: The Breakthrough Designation and Approval of Valbenazine". FDA. Archived from the original on 3 December 2020. Retrieved 15 November 2020.
- PMID 37783816.
- PMID 25063190.
- ISBN 978-0-19-851060-4.
- PMID 10434293.
- S2CID 20645822.
- ^ S2CID 46151626.
- ISBN 978-0-19-851060-4.
- PMID 8487273.
- ^ ISBN 978-0-19-851060-4.
- ^ Sharon I, Sharon R, Wilkens JP, Ersan T (2010). "Huntington Disease Dementia". emedicine, WebMD. Medscape. Archived from the original on 5 March 2010. Retrieved 16 May 2010.
- ISBN 978-0-323-03354-1.
- PMID 23482661.
- ISSN 0091-3952. NAID 10021247802.
- ^ S2CID 4320711.
- PMID 7881406.
- S2CID 207551998.
- PMID 25466405.
- S2CID 19786394.
- ^ ISBN 978-90-6186-011-2. Archivedfrom the original on 21 April 2022. Retrieved 21 April 2022.
- ^ Dunglison R (1842). The Practice of Medicine ... Vol. 2. Philadelphia, Pennsylvania, USA: Lea & Blanchard. pp. 312–313. Archived from the original on 20 April 2022. Retrieved 20 April 2022.
- ^ ISBN 978-0-19-851060-4.
- ^ Gorman CR (1846). On a Form of Chorea, Vulgarly called Magrums (Doctor of Medicine thesis). Philadelphia: Jefferson Medical College.
- "Catalogue of Jefferson Medical College of Philadelphia: Session of 1846-7". p. 14. Archived from the original on 26 June 2022.
- Dunglison R (1848). The Practice of Medicine ... Vol. 2 (3rd ed.). Philadelphia, Pennsylvania, USA: Lea & Blanchard. p. 218.
- Boyd WA (6 November 1913). "Hereditary chorea with report of a case". Boston Medical and Surgical Journal. 169 (19): 680–685. from the original on 2 August 2023. Retrieved 21 April 2022. See p. 680.
- ^ ISBN 978-0-300-10502-5. Retrieved 15 November 2015.
- ^ Lund JC (1860). "Chorea Sti Viti i Sætersdalen. Uddrag af Distriktslæge J. C. Lunds Medicinalberetning for 1860". Beretning Om Sundhedstilstanden: 137–138.
- S2CID 22659368.
- ^ Osler W (1908). "Historical notes on hereditary chorea". Neurographs. 1 (2): 113–116. Archived from the original on 21 April 2022. Retrieved 21 April 2022. See p. 115.
- See also: Osler W (February 1893). "Remarks on the varieties of chronic chorea, and a report upon two families of the hereditary form, with one autopsy". The Journal of Nervous and Mental Disease. 20 (2): 97–111. Archived from the original on 21 April 2022. Retrieved 21 April 2022. See p. 97.
- PMID 4228262.
- ^ Bateson W (1909). Mendel's Principles of Heredity. Cambridge, England: Cambridge University Press. p. 229. Bateson refers to "Huntington's disease" as "Hereditary Chorea".
- from the original on 5 April 2022. Retrieved 19 September 2020.
- ^ .
- from the original on 28 August 2021. Retrieved 1 April 2009.
- ^ S2CID 30791504.
- PMID 6233902.
- PMID 22248319.
- ^ a b "The Venezuela Huntington's disease project". Hereditary Disease Foundation website. Hereditary Disease Foundation. 2008. Archived from the original on 10 August 2015. Retrieved 8 September 2008.
- ^ S2CID 802885.
- PMID 15931380.
- S2CID 6603129.
- ^ S2CID 17488174.
- ^ a b Smith JE, Cordero C (23 May 2023). "Sought Out by Science, and Then Forgotten". The New York Times. Archived from the original on 23 May 2023. Retrieved 23 May 2023.
- ^ "What is HD?". Huntington's disease association. Archived from the original on 13 December 2011. Retrieved 18 December 2011.
- PMID 16575999.
- S2CID 18620094. Archived from the original(PDF) on 8 October 2020. Retrieved 1 December 2019.
- PMID 18181947.
- PMID 2140926.
- PMID 1971997.
- ^ "Insurance Genetics Moratorium extended to 2017" (Press release). Association of British Insurers. 5 April 2011. Archived from the original on 4 March 2016. Retrieved 13 January 2016.
- ^ "Expert backs gene test disclosure". BBC article. 7 June 2007. Archived from the original on 26 February 2008.
- PMID 8950670.
- PMID 18704981.
- ^ S2CID 39977672.
- ^ "The International Huntington Association". International Huntington Association. 2013. Archived from the original on 18 April 2009. Retrieved 3 April 2009.
- ^ "US Senate s. resolution 531". S. Res. 531. US Senate. 6 April 2008. Archived from the original on 17 November 2015. Retrieved 10 August 2008.
- ^ S2CID 4357517.
- ^ a b "Huntington's Disease: Hope Through Research". www.ninds.nih.gov. Archived from the original on 25 October 2020. Retrieved 16 November 2020.
- PMID 25155142.
- ^ a b Solomon S (3 May 2017). "Taube to fund $3m Huntington's disease research in US". The Times of Israel. Archived from the original on 3 May 2017. Retrieved 5 May 2017.
- ^ "Scientific Publications | CHDI Foundation". chdifoundation.org. Archived from the original on 12 December 2021. Retrieved 12 December 2021.
- ^ "CHDI Foundation". chdifoundation.org. Archived from the original on 14 November 2020. Retrieved 13 November 2020.
- S2CID 4318384.
- ^ PMID 21285520.
- PMID 22031240.
- PMID 22726834.
- PMID 3026739.
- ^ "Landmark Huntington's trial starts". Archived from the original on 21 October 2015. Retrieved 19 October 2015.
- ^ "Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of IONIS-HTTRx in Patients With Early Manifest Huntington's Disease - Full Text View". ClinicalTrials.gov. Archived from the original on 29 September 2015. Retrieved 18 April 2016.
- PMID 25844897.
- S2CID 38300571.
- PMID 26503961.
- S2CID 233173862.
- ^ Harding R (26 April 2021). Fox L (ed.). "Huntington's disease clinical trial round up". HDBuzz. Archived from the original on 5 May 2021. Retrieved 5 May 2021.
- ^ from the original on 23 May 2022. Retrieved 20 June 2023.
- S2CID 7749825.
- PMID 29512295.
- PMID 21277194.
- ^ PMID 28985560.
- ^ "Search of: Huntington Disease - List Results - ClinicalTrials.gov". clinicaltrials.gov. Archived from the original on 11 November 2021. Retrieved 16 November 2020.
- ^ "Completed Clinical Trials". Huntington Study Group. Archived from the original on 28 June 2012. Retrieved 4 February 2012.
External links
- Huntington's disease at Curlie
- HOPES project – Stanford University's HD information project
- HDBuzz – HD research news written by scientists in plain language
- HD Drug Works – news about current treatments and planned trials
