Atrop-abyssomicin C

| |
| Names | |
|---|---|
| IUPAC name
12,14a,3-(Epoxymethyno)-2H-1-benzoxacyclododecin-2,4,8(5H,10aH)-trione, 6,7,11,12,13,14-hexahydro-11-hydroxy-5,7,13-trimethyl-, (5R,7S,9E,10aR,11R,12R,13R,14aR)
| |
| Other names
Atrop-abyssomicin C
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C19H22O6 | |
| Molar mass | 346.38 g/mol |
| Density | 1.34±0.1 g/cm3 (Predicted) |
| Melting point | 180 °C (decomp) |
| Boiling point | 597.5±50.0 °C (Predicted) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Atrop-abyssomicin C is a polycyclic polyketide-type natural product that is the
Structure
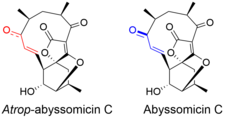
Atrop-abyssomicin C has a complex, yet intriguing structural topography. The compound contains an oxabicyclo[2.2.2]octane system fused to the tetronate moiety. The 11-membered macrocyclic ring carries an α,β-unsaturated ketone that was proposed to be the reactive center.[5] Despite being a strained macrocycle, there exist an atropisomer, abyssomicin C. The atropisomerism arise due to a structural deviation in the α,β-unsaturated ketone region of the molecule. The orientation of the carbonyl in atrop-abyssomicin C is cisoid, whereas the conformation in abyssomicin C is transoid.[6] The enone moiety of atrop-abyssomicin C has a higher degree of the conjugation, which makes it a more active Michael acceptor.[7]
Biosynthesis
The biosynthesis of atrop-abyssomicin C begins with the synthesis of a linear polyketide chain in a PKS I system that consist of one loading and six extension modules. The polyketide chain is made from five acetates, two propionates, and the glycolytic pathway metabolite. D-1,3-bisphosphoglycerate, the glycolytic metabolite, is transferred to AbyA3 (an acyl-carrier protein) by AbyA2 to generate the glyceryl-ACP. AbyA1 facilitates the attachment of the glyceryl-ACP to the polyketide chain and the detachment of the polyketide from the polyketide synthase to form intermediate 2.[7][8][9]

Based on the observation made for the biosynthesis of agglomerin, it has been proposed that AbyA4 acetylates intermediate 2 and AbyA5 catalyzes the elimination of acetic acid to form the exocyclic double bond in intermediate 4.[1] An intramolecular Diels-Alder was proposed to take place between the exocyclic olefin and the conjugated diene at the tail end of the polyketide to form the macrocyclic ring.[7] It has been reported that the previously unidentified Abycyc gene could code for an enzyme that carries out the Diels-Alder cycloaddition.[10] Following the Diels-Alder reaction, an epoxide ring is formed and then opened by the tetronate hydroxyl group to form atrop-abyssomicin C. It has been postulated that the AbyE monooxygenase catalyzes epoxide formation.[8]

