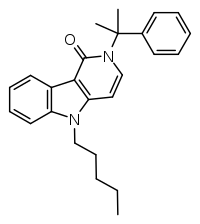
CUMYL-PEGACLONE
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
CUMYL-PEGACLONE (SGT-151) is a
Legal status
Sweden's public health agency classified CUMYL-PEGACLONE as a narcotic substance, on January 18, 2019.[12]
In the United States, the DEA has temporarily placed CUMYL-PEGACLONE into Schedule I status starting on December 12th, 2023 for up to 2 years, during which it's possible the DEA could file for permanent scheduling within those 2 years. If the DEA does not file for permanent placement the temporary Schedule I order will expire on December 12th, 2025.[13]
See also
- 5F-CUMYL-PEGACLONE
- CUMYL-CB-MEGACLONE
- CUMYL-CH-MEGACLONE
- CUMYL-BC-HPMEGACLONE-221
- CUMYL-PINACA
- CUMYL-5F-P7AICA
- UR-12
References
- ^ Anvisa (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.
- ^ "Substance Details CUMYL-PEGACLONE". Retrieved 2024-01-22.
- PMID 28601726.
- PMID 28670781.
- PMID 29314750.
- S2CID 53723006.
- S2CID 219285656.
- S2CID 226309264.
- ^ WO application 2001058869, Leftheris K, Zhao, R, Chen BC, Kiener P, Wu H, Pandit C, Chennagiri R, Wrobleski S, Chen P, Hynes j, Longphre M, Norris D, Spergel S, Tokarski J, "Cannabinoid Receptor Modulators, Their Processes of Preparation, and Use of Cannabinoid Receptor Modulators in Treating Respiratory and Non-Respiratory Diseases", published 16 August 2001, assigned to Bristol-Myers Squibb Company
- PMID 12747783.
- S2CID 209417068.
- ^ "Sexton nya ämnen klassas som narkotika eller hälsofarlig vara" (in Swedish). Folkhälsomyndigheten. 18 January 2019. Archived from the original on 3 June 2021. Retrieved 11 November 2019.
- ^ "Federal Register :: Request Access".
