Chanoclavine
| |
| Names | |
|---|---|
| IUPAC name
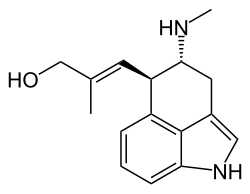
[9(9a)E]-9-Methyl-9,9a-didehydro-7,8-seco-9a-homoergolin-8-ol
| |
| Systematic IUPAC name
(2E)-2-Methyl-3-[(4R,5R)-4-(methylamino)-1,3,4,5-tetrahydrobenzo[cd]indol-5-yl]prop-2-en-1-ol | |
| Other names
chanoclavin-l
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C16H20N2O | |
| Molar mass | 256.34 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Chanoclavine, also known as chanoclavin-l is a tri-cyclic ergot alkaloid (
D2 receptors in the brain.[2]
See also
References
