Cilengitide
| |
| Names | |
|---|---|
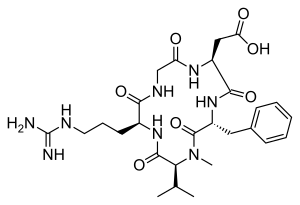
| IUPAC name
2-[(2S,5R,8S,11S)-5-benzyl-11-{3-[(diaminomethylidene)amino]propyl}-7-methyl-3,6,9,12,15-pentaoxo-8-(propan-2-yl)-1,4,7,10,13-pentaazacyclopentadecan-2-yl]acetic acid
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
IUPHAR/BPS |
|
| KEGG | |
| MeSH | Cilengitide |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C27H40N8O7 | |
| Molar mass | 588.656 g/mol |
| Density | 1.417 g/mL |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cilengitide (EMD 121974) is a molecule designed and synthesized at the
The European Medicines Agency has granted cilengitide orphan drug status.[3]
Cilengitide seems to function by inhibiting the
In a rat xenograft model, cilengitide was able to potentiate the cytotoxic effects of radiation when cilengitide was administered prior to radiation therapy.[5] When combined with radiation, inhibition of integrin expression by cilengitide synergistically improves the cytotoxic effects of ionizing radiation for glioblastoma.[5]
Clinical trials
Phase II studies were able to demonstrate that cilengitide as a potential
Cilengitide is well tolerated, in combination with radiation and temozolomide, at a dose of 2000 mg in patients with newly diagnosed glioblastoma, regardless of
However, in a subsequent study, cilengitide does not seem to alter the pattern of glioblastoma progression,[10] and in an EORTC phase III randomized, controlled, multicenter clinical trial, consisting of over 500 patients in 23 countries, the addition of cilengitide to the standard of care did not improve overall survival in patients with newly diagnosed glioblastoma and methylated MGMT promoter status [11] In 2014, a phase II study, the CORE trial, was conducted in patients with newly diagnosed glioblastoma and unmethylated MGMT promoter status.[12][needs update]
References
- PMID 12154028.
- PMID 11855984.
- ^ Spreitzer H (October 27, 2008). "Neue Wirkstoffe - Cilengitide". Österreichische Apothekerzeitung (in German) (22/2008): 1136–7.
- ^ S2CID 19861713.
- ^ S2CID 12073569.
- PMID 18981465.
- PMID 21739168.
- PMID 22517399.
- PMID 20439646.
- S2CID 21636884.
- ^ Merck Group. "Phase III Trial of Cilengitide Did Not Meet Primary Endpoint in Patients With Newly Diagnosed Glioblastoma, Date accessed: 3/24/2014."
- ^ ASCO Meeting Library. [1] "Cilengitide combined with standard treatment for patients with newly diagnosed glioblastoma and methylated O6-methylguanine-DNA methyltransferase (MGMT) gene promoter: Key results of the multicenter, randomized, open-label, controlled, phase III CENTRIC study, Date accessed: 3/24/2014."
