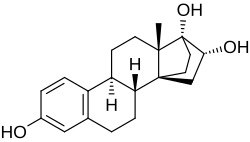
Cyclotriol
 | |
| Clinical data | |
|---|---|
| Other names | ZK-136295; Cycloestriol; 14α,17α-Ethanoestriol; 14α,17α-Ethanoestra-1,3,5(10)-triene-3,16α,17β-triol; 14,21-Cyclo-19-norpregna-1,3,5(10)-triene-3,16α,17α-triol |
| Routes of administration | By mouth[1] |
| Drug class | Estrogen |
| Pharmacokinetic data | |
| Bioavailability | 40%[1] |
| Elimination half-life | 12.3 hours[1] |
| Identifiers | |
| |
JSmol) | |
| |
| |
Cyclotriol (developmental code name ZK-136295; also known as 14α,17α-ethanoestriol) is a
pharmacokinetic studies in women.[1]
See also
References
- ^ PMID 8922877.
- ^ ISBN 978-3-642-60107-1.
- ^ S2CID 39049586.
- S2CID 10326219.
- PMID 9093381.
