Dicarbollide
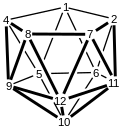 Numbering system for nido-11 vertex clusters.
| |
| Identifiers | |
|---|---|
3D model (
JSmol ) |
|
| |
| |
| Properties | |
| C2H11B9− | |
| Molar mass | 132.40 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
In
Synthesis of dicarbollides
Dicarbollides are obtained by base-degradation of 12-vertex dicarboranes. This degradation of the ortho derivative has been most heavily studied. The conversion is conducted in two-steps, first "deboronation" and second deprotonation:[2]

- C2B10H12 + NaOEt + 2 EtOH → Na+C2B9H12− + H2 + B(OEt)3
- Na+C2B9H12− + NaH → Na2C2B9H11 + H2
The dianion derived from dicarboranes, [C2B9H11]2-, are nido clusters. Three isomers exist. Most commonly studies is the 7,8-isomer, with two adjacent carbon centers on the rim. 7,9-C2B9H2−11 has non-adjacent carbon centers on the rim. It is derived by degradation of meta-C2B10H12. 2,9-C2B9H2−11 has only one carbon center on the rim. It is derived by degradation of para-C2B10H12.[3]
Coordination compounds

A variety of complexes - a subset of metallaborane - are known with one or two dicarbollide ligands. An example of a 1:1 complex is [Mn(CO)3(η5-7,8-C2B9H11)]−.[5]
Most heavily studied are complexes with two dicarbollide ligands, especially
- 2 Na2C2B8H11 + FeCl2 → Na2[Fe(C2B8H11)2] + 2 NaCl
These bisdicarbollide dianions are often readily oxidized. Fe(III), Co(III), Ni(III), and Ni(IV) derivatives are known. In some cases, the oxidation induces rearrangement of the C2B9 cage to give complexes where the carbon centers are nonadjacent.[1]
Precursor to other carboranes
Diprotonation of [C2B9H11]2− gives the neutral carborane C2B9H13. Pyrolysis of this nido cluster gives closo-C2B9H11. Chromate-oxidation of [C2B9H12]− results in deboronation, giving the C2B7H13. This carborane features two CH2 vertices.[6]
Homogeneous catalysis
The clam-shell dicarbollide complex (Cp*)(C2B9H11)ZrCH3 catalyzes alkene polymerization.[7]
References
- ^ .
- ISBN 978-0-470-13253-1.
- doi:10.1039/B108937D.
- .
- .
- ISBN 978-0-12-801894-1.
- .
