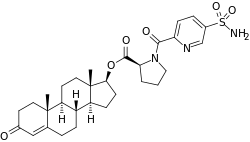
EC586
Appearance
 | |
 | |
| Clinical data | |
|---|---|
| Other names | EC-586; Testosterone 17β-(1-[[5-(aminosulfonyl)-2-pyridinyl]carbonyl]-L-proline); Androst-4-en-17β-ol-3-one 17β-(1-[[5-(aminosulfonyl)-2-pyridinyl]carbonyl]-L-proline); 3-Oxoandrost-4-en-17β-yl 1-[[5-(aminosulfonyl)-2-pyridinyl]carbonyl]-L-proline |
| Drug class | Androgen; Anabolic steroid; Androgen ester |
| Identifiers | |
| |
JSmol) | |
| |
| |
EC586, also known as testosterone 17β-(1-((5-(aminosulfonyl)-2-pyridinyl)carbonyl)-L-proline), is an
parenteral testosterone, but with the convenience of oral administration.[2][3] Evestra intends to seek Investigational New Drug status for EC586 in the fourth quarter of 2018.[needs update][1]
The
propionate ester of testosterone (AUC0-3h = 330 ng/mL and 2.5 ng/mL, respectively, for doses of 3.0 mg/rat each).[2] As such, EC586 would appear to possess strongly increased oral bioavailability, potency, and systemic exposure relative to testosterone propionate.[2] Additional research and details on the pharmacokinetics and properties of EC586 are to be published "soon".[2]
The mechanism for the absence of first-pass metabolism and lack of disproportionate liver exposure with oral administration has been elucidated for a closely related sulfonamide–proline
Clinical trials for EC586 and EC508 are undergoing as of 2023.[4]
See also
- List of androgen esters § Testosterone esters
- List of investigational sex-hormonal agents § Androgenics
References
- ^ a b "Research Pipeline". Evestra, Inc. Archived from the original on 4 March 2018.
EC586: Testosterone prodrug
- ^ PMID 28886996.
- ^ S2CID 26650319.
- S2CID 259299284.
External links
- R&D Research / Research Pipeline - Evestra, Inc. Archived 2017-09-29 at the Wayback Machine
