Europium anomaly
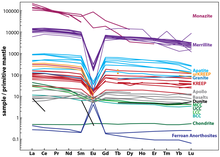
The europium anomaly is the phenomenon whereby the
While all lanthanides form relatively large trivalent (3+) ions, Eu and cerium (Ce) have additional valences, europium forms 2+ ions, and Ce forms 4+ ions, leading to chemical reaction differences in how these ions can partition versus the 3+ REEs. In the case of Eu, its reduced divalent (2+) cations are similar in size and carry the same charge as Ca2+, an ion found in plagioclase and other minerals. While Eu is an incompatible element in its trivalent form (Eu3+) in an oxidizing magma, it is preferentially incorporated into plagioclase in its divalent form (Eu2+) in a reducing magma, where it substitutes for calcium (Ca2+).[2]
Enrichment or depletion is generally attributed to europium's tendency to be incorporated into plagioclase preferentially over other minerals. If a magma crystallizes stable plagioclase, most of the Eu will be incorporated into this mineral, causing a higher than expected concentration of Eu in the mineral versus other REE in that mineral (a positive anomaly). The rest of the magma will then be relatively depleted in Eu with a concentration of Eu lower than expected versus the concentrations of other REEs in that magma. If the Eu-depleted magma is then separated from its plagioclase crystals and subsequently solidifies, its chemical composition will display a negative Eu anomaly (because the Eu is locked up in the plagioclase left in the magma chamber). Conversely, if a magma accumulates plagioclase crystals before solidification, its rock composition will display a relatively positive Eu anomaly.[3][4]
A well-known example of the Eu anomaly is seen on the Moon. REE analyses of the Moon's light-colored
See also
References
- doi:10.3390/resources6030040.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - ISBN 978-90-277-1613-2.
- PMID 17806582.
- .
- ISBN 978-0-521-87862-3.
