Fischer oxazole synthesis
| Fischer oxazole synthesis | |
|---|---|
| Named after | Hermann Emil Fischer
|
| Reaction type | Ring forming reaction |
The Fischer oxazole synthesis is a
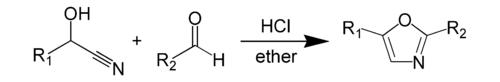
A more specific example of Fischer oxazole synthesis involves reacting mandelic acid nitrile with benzaldehyde to give 2,5-diphenyl-oxazole.[4]
History
Fischer developed the Fischer oxazole synthesis during his time at Berlin University. The Fischer oxazole synthesis was one of the first syntheses developed to produce 2,5-disubstituted oxazoles.[4]
Mechanism
The Fischer oxazole synthesis is a type of
The
Applications
Diarylazoles are common
The Fischer oxazole synthesis has also been useful in the synthesis of 2-(4-Bromophenyl)5-phenyloxazole starting with benzaldehyde cyanohydrin and 4-bromobenzaldehyde. However, oxazole ring chlorination occurs to give 2,5-bis(4-bromophenyl)-4-chlorooxazole 7 along with 2,5-bis(4-bromophenyl)-4-oxazolidinone 8. The latter compound is in general a by-product.[6]

Another useful example is the one pot two-step synthesis of halfordinol, a parent compound for Rutaceae alkaloids. The initial steps follow the Fischer oxazole synthesis, although the acid-catalyzed cyclization occurs in two steps rather than one, which ensures the formation of the di-chloro intermediate, preventing formation of the regioisomer.[4]

In recent research,[citation needed] a reconsideration of the Fischer oxazole synthesis has led to the synthesis of 2,5-disubstituted oxazoles from aldehydes and α-hydroxy-amides. However, unlike the Fischer oxazole synthesis, the new method is not limited to diaryloxazoles.[7]
References
- ^ )
- Fischer, E. Ber.1896, 29, 205.
- ^ Li, J. J. Fischer Oxazole Synthesis. In Name Reactions: A Collection of Detailed Mechanisms and Synthetic Applications, 4th ed.; Springer-Verlag Berlin Heidelberg: New York, 2003, 229-230. (Review). ([1] Archived 2016-03-04 at the Wayback Machine)
- ^ a b c d Maklad, N. Name Reactions in Heterocyclic Chemistry II; Li, J.J.; Wiley & Sons; Hoboken, NJ, 2011, 225-232. ([2])
- )
- ^ Turchi, I. J. Oxazole Chemistry: A Review of Recent Advances. Ind. Eng. Che. Prod. Res. Dev. 1981, 20, 32-76. ([3]) (Review).
- )
