Fluorescamine
Appearance
| |
| Names | |
|---|---|
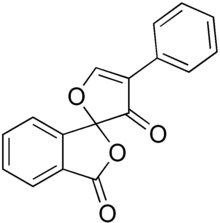
| IUPAC name
4'-phenylspiro[2-benzofuran-3,2'-furan]-1,3'-dione
| |
| Other names
Fluram
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.048.904 |
| MeSH | D005450 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C17H10O4 | |
| Molar mass | 278.26 g/mol |
| Melting point | 153 to 157 °C (307 to 315 °F; 426 to 430 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Fluorescamine is a spiro compound that is not fluorescent itself, but reacts with primary amines to form highly fluorescent products, i.e. it is fluorogenic. It hence has been used as a reagent for the detection of amines and peptides.[2] 1-100 μg of protein and down to 10 pg of protein can be detected.[3][4] Once bound to protein the excitation wavelength is 381 nm (near ultraviolet) and the emission wavelength is 470 nm (blue).[5] This method is found to suffer from high blanks resulting from a high rate of hydrolysis due to requiring a large excess concentration.[6] Alternative methods are based on ortho-phthalaldehyde (OPA), Ellman's reagent (DTNB), or epicocconone.
Reaction
See also
References
- ^ Fluram at Sigma-Aldrich
- .
- PMID 4736505.
- Fluoprobes
- ^ Biotium. "Fluorescamine PRODUCT AND SAFETY DATA SHEET" (PDF). Biotium. Retrieved 21 February 2023.
- ^ [1]

