Guerbet reaction
The Guerbet reaction, named after Marcel Guerbet (1861–1938), is an
dimer alcohol with loss of one equivalent of water. The process is of interest because it converts simple inexpensive feedstocks into more valuable products. Its main disadvantage is that the reaction produces mixtures.[1]

Scope and applications
The original 1899 publication concerned the conversion of
2-ethylhexanol.[2] 2-ethylhexanol is however more easily prepared by alternative methods (from butyraldehyde
).
Instead, the Guerbet reaction is mainly applied to
Raney Nickel are required to facilitate the hydrogen transfer steps.[1]
While the Guerbet reaction is traditionally (and commercially) focused on fatty alcohols, it has been investigated for the dimerization of ethanol to butanol.[3]
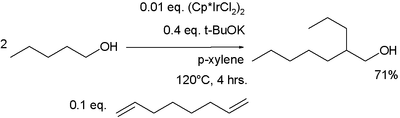
Organometallic catalysts have been investigated.1,7-octadiene is required as a proton acceptor.
Mechanism
The
oxidized to the aldehyde. These intermediates then react in an aldol condensation to the allyl aldehyde which the hydrogenation catalyst then reduces to the alcohol.[5]

The Cannizzaro reaction is a competing reaction when two aldehyde molecules react by disproportionation to form the corresponding alcohol and carboxylic acid. Another side reaction is the Tishchenko reaction.
See also
- Oxo alcohols - a different reaction which gives similar products
- Guerbet alcohols
- 2-Ethyl-1-butanol
- 2-Ethylhexanol
- 2-Propylheptan-1-ol
- 2-Butyl-1-octanol
- 2-Butyl-1-octanol
References
- ^ ISBN 978-3527306732.
- ^
Marcel Guerbet (1909). "Condensation de l'alcool isopropylique avec son dérivé sodé; formation du méthylisobutylcarbinol et du diméthyl-2.4-heptanol-6". Comptes rendus de l'Académie des sciences. 149: 129–132.
- .
- PMID 17025333.
- .
External links
- A Review of Guerbet Chemistry Anthony J. O’Lenick, Jr. https://web.archive.org/web/20110209074739/http://www.zenitech.com/ Link
