Hydroacylation
Hydroacylation is a type of
unsaturated hydrocarbon inserts into a formyl C-H bond. With alkenes, the product is a ketone
:
- RCHO + CH2=CHR' → RC(O)CH2CH2R'
With an alkyne instead, the reaction produces an
α,β-unsaturated ketone.[2]
The reaction requires a metal
homogeneous catalysts
, often based on rhodium phosphines.
History
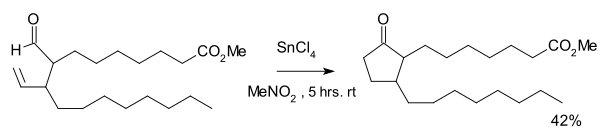
The reaction was discovered in the 1970s as part of a synthetic route to certain
tin tetrachloride and a stoichiometric amount of Wilkinson's catalyst
:
An equal amount of a cyclopropane was formed as the result of decarbonylation.
The first
catalytic application involved cyclization of 4-pentenal to cyclopentanone using (again) Wilkinson's catalyst.[4] In this reaction the solvent was saturated with ethylene
.
- CH2=CHCH2CH2CHO → (CH2)4CO
Reaction mechanism
Labeling studies establish the following regiochemistry:
- RCDO + CH2=CHR' → RC(O)CH2CHDR'
In terms of the
metal hydride
:
- R"C(O)-MLn-H → R"-M(CO)Ln-H
This step can be followed by reductive elimination of the alkane:
Asymmetric hydroacylation
Hydroacylation as an