Iodite
(Redirected from
Iodous acid
)
| |
| Names | |
|---|---|
| IUPAC name
iodite
| |
| Systematic IUPAC name
dioxidoiodate(1−) | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| IO− 2 | |
| Molar mass | 58.90 g/mol |
Conjugate acid
|
Iodous acid
|
| Related compounds | |
Other anions
|
Bromite
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
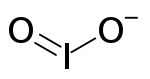
The iodite
anion, is the halite with the chemical formula IO−
2. Within the ion the Iodine exists in the oxidation state
of +3.
2. Within the ion the Iodine exists in the oxidation state
Iodite anion
Iodites (including
iodous acid) are highly unstable and have been observed[1] but never isolated.[citation needed] They will rapidly disproportionate to molecular Iodine and Iodates.[2] However, they have been detected as intermediates in the conversion between iodide and iodate.[3][4]
Iodous acid

| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
iodous acid
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChemSpider | |||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| HIO2 | |||
| Molar mass | 159.91 g/mol | ||
Conjugate base
|
Iodite | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Iodous acid is acid form of the iodite ion, with the formula HIO2.
Other oxyanions
Iodine can assume oxidation states of −1, +1, +3, +5, or +7. A number of neutral iodine oxides are also known.
| Iodine oxidation state | −1 | +1 | +3 | +5 | +7 |
|---|---|---|---|---|---|
| Name | Iodide | Hypoiodite
|
Iodite | Iodate | periodate |
| Formula | I− | IO− | IO− 2 |
IO− 3 |
IO− 4 or IO5− 6 |
References
- ISBN 978-0-08-037941-8.
- ISBN 978-0-08-037941-8.
- .
- doi:10.1063/1.462352.


