Periodate
 The metaperiodate ion
| |
 The orthoperiodate ion
| |
| Names | |
|---|---|
| Systematic IUPAC name
tetraoxoiodate(1−) hexaoxoiodate(5−) | |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| UNII |
|
| |
| |
| Properties | |
| IO4− or IO65- | |
Conjugate acid
|
Periodic acid |
| Related compounds | |
Other anions
|
Perchlorate Perbromate Permanganate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Periodate (
Periodates were discovered by Heinrich Gustav Magnus and C. F. Ammermüller; who first synthesised periodic acid in 1833.[1]
Synthesis
Classically, periodate was most commonly produced in the form of sodium hydrogen periodate (Na3H2IO6).[2] This is commercially available, but can also be produced by the oxidation of iodates with chlorine and sodium hydroxide.[3] Or, similarly, from iodides by oxidation with bromine and sodium hydroxide:
Modern industrial scale production involves the
Metaperiodates are typically prepared by the dehydration of sodium hydrogen periodate with nitric acid,[2] or by dehydrating orthoperiodic acid by heating it to 100 °C under vacuum.
They can also be generated directly from iodates by treatment with other strong oxidizing agents such as hypochlorites:
Forms and interconversion
Periodate can exist in a variety of forms in aqueous media, with pH being a controlling factor. Orthoperiodate has a number of acid dissociation constants.[5][6]
- pKa = 3.29
- pKa = 8.31
- pKa = 11.60
The ortho- and metaperiodate forms also exist in equilibrium.
- K = 29
For this reason orthoperiodate is sometimes referred to as the dihydrate of metaperiodate,[7] written IO−4·2H2O; however, this description is not strictly accurate as X-ray crystallography of H5IO6 shows 5 equivalent I−OH groups.[8]
At extremes of pH additional species can form. Under basic conditions a dehydration reaction can take place to form the diperiodate (sometimes referred to as mesoperiodate).
- K = 820
Under strongly acid conditions periodic acid can be protonated to give the orthoperiodonium cation.[9]
- pKa = −0.8
Structure and bonding
In both the ortho- and metaperiodate the iodine is
Exact structures vary depending on counter ions, however on average orthoperiodates adopt a slightly deformed octahedral geometry with
Reactions
Cleavage reactions
Periodates can cleave carbon–carbon bonds on a variety of 1,2-difunctionalised alkanes.

Alkenes can also be oxidised and cleaved in the Lemieux–Johnson oxidation. This uses a catalytic loading of osmium tetroxide which is regenerated in situ by the periodate. The overall process is equivalent to that of ozonolysis.
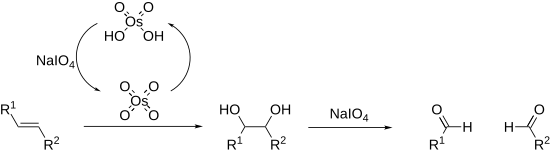
Cleavage reactions proceed via a cyclic intermediate called a periodate ester. The formation of this may be affected by pH and temperature

Periodate cleavage is often utilized in molecular biochemistry for the purposes of modifying
Periodate cleavage may be performed on an industrial scale to form dialdehyde starch which has uses in paper production.[26]
Oxidation reactions
Periodates are powerful
Niche uses
Periodates are highly selective etchants for certain ruthenium-based oxides.[31]
Several staining agents use in microscopy are based around periodate (e.g. periodic acid–Schiff stain and Jones' stain).
Periodates have also been used as oxidising agents for use in
Other oxyanions
Periodate is part of a series of oxyanions in which iodine can assume oxidation states of −1, +1, +3, +5, or +7. A number of neutral iodine oxides are also known.
| Iodine oxidation state | −1 | +1 | +3 | +5 | +7 |
|---|---|---|---|---|---|
| Name | iodide | hypoiodite
|
iodite | iodate | periodate |
| Formula | I− | IO− | IO− 2 |
IO− 3 |
IO− 4 or IO5− 6 |
| Structure | 
|
See also
References
- .
- ^ ISBN 012126601X.
- .
- ^ Parsons, Roger (1959). Handbook of electrochemical constants. Butterworths Scientific Publications Ltd. p. 71.
- ISBN 0123526515.)
{{cite book}}: CS1 maint: multiple names: authors list (link - ISBN 978-1441983824.
- ISBN 978-0444595539.
- ^ .
- ISBN 0750633654.
- PMID 25045143.
- .
- .
- .
- .
- ISBN 9780080868011.
- Bull. Soc. Chim. Fr.3, 1, 833 (1934)
- PMID 14342227.
- .
- .
- S2CID 97403497.
- .
- .
- ISBN 978-0840054531.
- .
- ISBN 0387949518.
- .
- .
- .
- doi:10.1139/v55-208.
- ISSN 0022-3263.
- ^ Dieter Weber, Róza Vöfély, Yuehua Chen, Yulia Mourzina, Ulrich Poppe: Variable resistor made by repeated steps of epitaxial deposition and lithographic structuring of oxide layers by using wet chemical etchants. Thin Solid Films (2013) DOI: 10.1016/j.tsf.2012.11.118
- PMID 22639415.
- ^ "Picatinny to remove tons of toxins from lethal rounds". U.S. Army. Retrieved 31 October 2013.












