Jablonski diagram

This article needs additional citations for verification. (July 2023) |
In molecular
Transitions
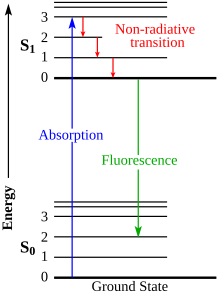
When a molecule absorbs a photon, the photon energy is converted and increases the molecule's internal energy level. Likewise, when an excited molecule releases energy, it can do so in the form of a photon. Depending on the energy of the photon, this could correspond to a change in vibrational, electronic, or rotational energy levels. The changes between these levels are called "transitions" and are plotted on the Jablonski diagram.
Radiative transitions involve either the absorption or emission of a photon. As mentioned above, these transitions are denoted with solid arrows with their tails at the initial energy level and their tips at the final energy level.
Nonradiative transitions arise through several different mechanisms, all differently labeled in the diagram. Relaxation of the excited state to its lowest vibrational level is called vibrational relaxation. This process involves the dissipation of energy from the molecule to its surroundings, and thus it cannot occur for isolated molecules.
A second type of nonradiative transition is internal conversion (IC), which occurs when a vibrational state of an electronically excited state can couple to a vibrational state of a lower electronic state. The molecule could then subsequently relax further through vibrational relaxation.[4]
A third type is
See also
- Franck–Condon principle
- Grotrian diagram (for atoms)
References
- ^ “Jablonski Diagram.” 2006. In IUPAC Compendium of Chemical Terminoloy, 3rd ed. International Union of Pure and Applied Chemistry. https://doi.org/10.1351/goldbook.J03360.
- ISBN 0-7167-8759-8
- doi:10.1038/131839b0
- ^ Harris, D. C. Lucy, C. A. Quantitative Chemical Analysis, Tenth Edition (2020), pp 457-458, W.H. Freeman and Co.
External links
- Florida State University: Jablonski diagram primer
- Consequences of Light Absorption – The Jablonski Diagram
