Mono-N-protected amino acids
Mono-N-protected amino acid (MPAA) is a bifunctional ligand that plays a key role in C–H functionalizations by accelerating the reaction rate and imparting specified chirality into the product.[1] Amino acids are ideal building blocks for chiral ligand synthesis due to the cost, accessibility, large variety, solubility, and inherent chirality.[2] Naturally occurring amino acids are transformed into chiral MPAA ligands that, upon coordination to metal complexes, allow reactions to occur that are otherwise energetically unfavorable. Great strides in the development of MPAA ligands over the past two decades have led to the integral role that enantioselective catalysis now plays in complex organic synthesis.[3]
History and development
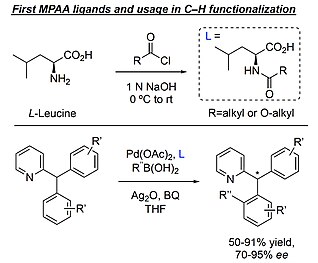
In the past century, there has been much research into the development of effective chiral catalysts due to its great potential in organic synthesis.[4] In the 1960s, cyclometalation reactions including C(sp2)–H and C(sp3)–H cleavage were pioneered by Kleiman, Dubeck, Cope, and Siekman.[5][6] A decade later, Shaw discovered that inorganic acetate salts promoted otherwise difficult cyclopalladations.[7] To build off of this work, Sokolov focused on developing chiral, enantioenriched metallacyles and proposed the concerted metal-deprotonation (CMD) mechanism.[8]
Despite this foundation of discoveries, enantioselective catalysis for C–H functionalization continued to lack in efficiency oand selectivity for desired chiral product formation.[3] In 2008, Jin-Quan Yu reported the first MPAA ligands, showcasing their use in enantioselective activation of C(sp2)–H and C(sp3)–H bonds.[9] Initial synthesis occurred by reacting the nucleophilic amino acid in base with a highly electrophilic acyl chloride resulting in one new amide bond formation. Upon addition of acyl chloride, most resulting groups off of the nitrogen were common protecting groups used in organic synthesis, hence mono-N-protected. Taking advantage of the weak coordination of amides and carboxylates with Pd-complexes, this enantioselective catalysis requires the MPAA ligand to allow the reaction to proceed and determine the product chirality, minimizing side reactions that may occur without the ligand.[1] Since the initial discovery, Yu has continued to pioneer the field by expanding the substrate scope, increasing functional group tolerance, and developing ligand variations.
Mechanism
After iterative computational and experimental studies, the internal amidate mechanism was proposed in collaboration of Wu,

The key intermediate step of these cyclometalation reactions involves the metal-mediated cleavage of the C–H bond and simultaneous formation of the metal–C bond of the substrate. Upon addition of substrate, the N-acyl motif acts as an internal proton acceptor in the concerted metal-deprotonation (CMD) of the transition state for this inner-sphere process. According to this model, the rate and selectivity of the C–H functionalization are impacted by the basicity of the MPAA ligand. The resulting experimental data of steric and electronic alteration of the MPAA ligands align with this model.[12] In 2023, the first experimental observations to support the proposed mechanism were reported, which were previously unattainable due to the lack of well-defined isolated palladium-MPAA complexes.[13]
Ligand variations
Since the initial MPAA ligand report, many variations of bifunctional ligands derived from amino acids have been developed. Bidentate MPAQ (mono-protected amino
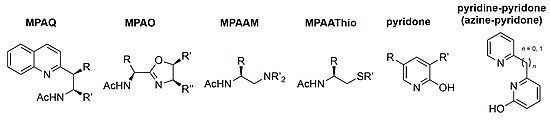
Expanding the reaction substrate scope to non-directed C(sp2)–H bonds, pyridone ligands were developed to functionalize arenes and heteroarenes which proved to be particularly useful in late-state derivatization of bioactive compounds such as estrone, caffeine, and camptothecin.[22][23] Many analogues of the pyridine-pyridone (azine-pyridone) ligands were developed and used in the C(sp2)–H hydroxylation of (hetero)arenes and the dehydrogenation of methylene C(sp3)–H bonds on alkyl free acids.[24][25]
Applications in total synthesis

The development of MPAA ligands enabled and improved the synthesis of many complex natural products. Examples include Arnottin 1,[26] Aspercylide B,[27] Berkelic Acid,[28] Boletopsin 11,[29] Danshenspiroketallactone,[29] Delavatine A,[30] Herbindole B/cis-Trikentrin A,[31] Hongoquercin A,[32] Incarviatone A,[33] Indoxamycin,[34] Kedarcidin/Neocarzinostatin,[35] Kinamycin[36]
, Lithospermic Acid,[37] M1 PAMs,[38] and VS-548.[39] In the formation of indoxamycin cores, MPAA ligand assisted C–H functionalization introduces high complexity via intramolecular ortho olefination.[34]
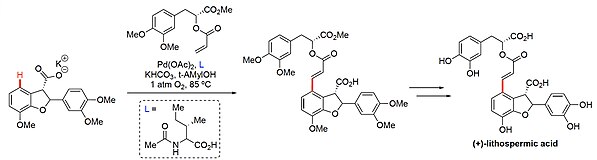
The use of C–H functionalization in the synthesis of lithospheric acid exemplifies the site- and stereoselective capabilities of using MPAA ligands in these reactions. As the penultimate step, the intermolecular C–H olefination introduces almost double the complexity into the compound enabling the highly convergent synthesis.[37]
References
- ^ ISSN 1365-3075.
- ISBN 978-3-527-30385-4.
- ^ PMID 32227915.
- ISSN 0001-4842.
- ISSN 0002-7863.
- ISSN 0002-7863.
- ISSN 0022-328X.
- ISSN 0022-328X.
- PMID 18484582.
- PMID 22148424.
- PMID 24410499.
- ISSN 0276-7333.
- S2CID 258943376.
- PMID 27701111.
- PMID 28154075.
- PMID 28209055.
- PMID 29531850.
- PMID 29629766.
- PMID 29741883.
- PMID 30029574.
- PMID 32605367.
- PMID 27712063.
- PMID 29168802.
- PMID 34840353.
- PMID 34762490.
- PMID 21405112.
- PMID 19418521.
- S2CID 228922066.
- ^ PMID 29792705.
- PMID 28271887.
- S2CID 7847026.
- PMID 23740529.
- PMID 26371964.
- ^ PMID 27206223.
- PMID 19965380.
- .
- ^ PMID 21443224.
- PMID 28598634.
- PMID 26841330.
This article needs additional or more specific categories. (June 2023) |
