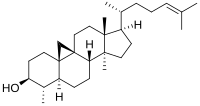
Norcycloartenol
Appearance
| |
| Names | |
|---|---|
| Other names
4α,14α-dimethyl-9β,19-cyclo-5α-cholest-24-en-3β-ol
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C29H48O | |
| Molar mass | 412.702 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
29- or 31-Norcycloartenolsterol biosynthesis.[1][2][3][4] In the pathway, it is transformed from demethylation of cycloartenol, then 9,19-cyclopropyl-ring opening reaction occurs to 29-Norlanosterol.[5]
Note
- ^ zoology and botany are different in numbering of steroids side chain, 241 to 28, 242 to 29
