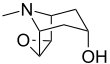
Scopine
Appearance
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
6β,7β-Epoxytropan-3α-ol
| |||
| Systematic IUPAC name
(1R,2R,4S,5S,7s)-9-Methyl-3-oxa-9-azatricyclo[3.3.1.02,4]nonan-7-ol | |||
| Other names
6,7-Epoxytropine; Scopanol; Scopin; 6β,7β-Epoxy-1αH,5αH-tropan-3α-ol
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEMBL | |||
| ChemSpider | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C8H13NO2 | |||
| Molar mass | 155.197 g·mol−1 | ||
| Melting point | 75 to 76 °C (167 to 169 °F; 348 to 349 K)[1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
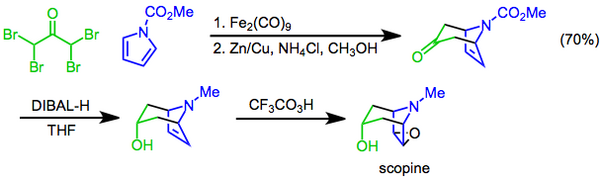
Scopine is a
Scopine can be prepared by the
diisobutylaluminum hydride, and finally a Prilezhaev epoxidation with trifluoroperacetic acid affords scopine.[8]
See also
References
- ^ PMID 6044210.
- .
- .
- ^ Bendik, I.; Bauerova, O.; Bauer, S.; Mokry, J.; Tomko, J. (1958). "Alkaloids from Scopolia carniolica". Chemicke Zvesti. 12: 181–184.
- ^ Szymanska, Miroslawa (1967). "Alkaloids in Scopolia lurida. Chromatographic analysis. Isolation of cuscohygrine". Acta Poloniae Pharmaceutica. 24 (1): 59–64.
- .
- .
- .