Tetrabenzylzirconium
Appearance

| |
| Identifiers | |
|---|---|
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C28H28Zr | |
| Molar mass | 455.756 g·mol−1 |
| Appearance | orange solid |
| Density | 1.34-1.39 g/cm3[1] |
| Melting point | 133–134 °C (271–273 °F; 406–407 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tetrabenzylzirconium is an
benzyl ligands. It is an orange air- and photo-sensitive solid, which is soluble in hydrocarbon solvents. The compound is a precursor to catalysts for the polymerization of olefins.[2]
[3]
Structure, synthesis, reactions
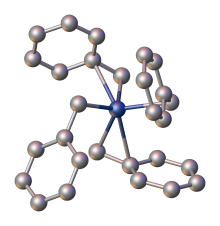
The compound is prepared by combining benzylmagnesium chloride and
zirconium tetrachloride in diethyl ether.[4]
Tetrabenzylzirconium readily undergoes protonolysis, e.g. with hydrogen chloride:
- Zr(CH2C6H5)4 + HCl → Zr(CH2C6H5)3Cl + CH3C6H5
References
- ^ .
- .
- .
- ISBN 978-0471936237.
