Thorpe reaction
The Thorpe reaction is a
aliphatic nitriles catalyzed by base to form enamines.[1][2][3] The reaction was discovered by Jocelyn Field Thorpe
.
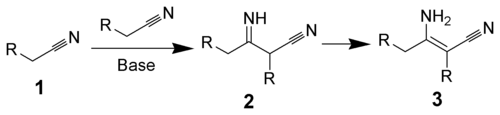
Thorpe–Ziegler reaction
The Thorpe–Ziegler reaction (named after Jocelyn Field Thorpe and Karl Ziegler), or Ziegler method, is the intramolecular modification with a dinitrile as a reactant and a cyclic ketone as the final reaction product after acidic hydrolysis. The reaction is conceptually related to the Dieckmann condensation.[3]

References
- ISSN 0368-1645.
- ISSN 0075-4617.
- ^ ISBN 978-0471264187.
External links
- Thorpe-Ziegler reaction: 4-Phosphorinanone, 1-phenyl- Organic Syntheses, Coll. Vol. 6, p. 932 (1988); Vol. 53, p. 98 (1973) Link
