Tonicity
This article needs additional citations for verification. (February 2018) |
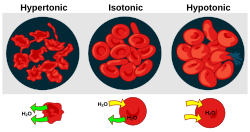

In
Unlike osmotic pressure, tonicity is influenced only by solutes that cannot cross the membrane, as only these exert an effective osmotic pressure. Solutes able to freely cross the membrane do not affect tonicity because they will always equilibrate with equal concentrations on both sides of the membrane without net solvent movement. It is also a factor affecting imbibition.
There are three classifications of tonicity that one solution can have relative to another: hypertonic, hypotonic, and isotonic.[1] A hypotonic solution example is distilled water.
Hypertonic solution

A hypertonic solution has a greater concentration of non-permeating
When plant cells are in a hypertonic solution, the flexible cell membrane pulls away from the rigid
Some organisms have evolved intricate methods of circumventing hypertonicity. For example, saltwater is hypertonic to the fish that live in it. Because the fish need a large surface area in their gills in contact with seawater for gas exchange, they lose water osmotically to the sea from gill cells. They respond to the loss by drinking large amounts of saltwater, and actively excreting the excess salt.[6] This process is called osmoregulation.[7]
Hypotonic solution

A hypotonic solution has a lower concentration of solutes than another solution. In biology, a solution outside of a cell is called hypotonic if it has a lower concentration of solutes relative to the
Isotonicity

A solution is isotonic when its effective
An iso-osmolar solution can be hypotonic if the solute is able to penetrate the cell membrane. For example, an iso-osmolar
See also
References
- ISBN 978-0-12-387738-3.
- ^ Buckley, Gabe (20 January 2017). "Hypertonic Solution". In Biologydictionary.net (ed.). Biology Dictionary (Online ed.). Biologydictionary.net. Retrieved 19 August 2021.
- ^ LibreTexts Project: Medicine (18 July 2018). "3.3C - Tonicity". Anatomy and Physiology (Boundless) (Online ed.). med.libretexts.org/. Retrieved 19 August 2021.
- PMID 27382523.
- ^ Lodish, Harvey; Berk, Arnold; Zipursky, S. Lawrence; Matsudaira, Paul; Baltimore, David; Darnell, James (2000). "Osmosis, Water Channels, and the Regulation of Cell Volume". Molecular Cell Biology (4th ed.). New York: W. H. Freeman and Company. Retrieved 19 August 2021.
- ^ Soult, Allison (2020). "8.4 - Osmosis and Diffusion". In University of Kentucky (ed.). Chemistry for Allied Health. Open Education Resource (OER) LibreTexts Project. Retrieved 19 August 2021.
- PMID 11441026.
- ^ "Definition — hypotonic". The Free Dictionary. Retrieved 23 August 2012.
