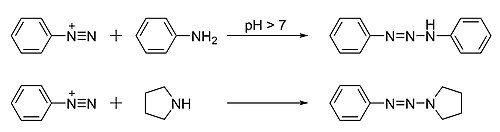
Triazenes
Triazenes are organic compounds that contain the functional group R1−N=N−NR2R3, where the R are each any of various types of substituent groups.[2] Some anti-cancer medications and dyes are triazenes.[3] Formally, the triazenes are related to the unstable chemical triazene, H2N−N=NH.
Production
Triazenes are prepared from the N-coupling reaction between
The diazonium reagents are themselves available starting from amines. For symmetrical triazenes derived from primary amines, partial diazotization gives a mixture of the original amine and its diazo derivative that then couple with each other. For example,
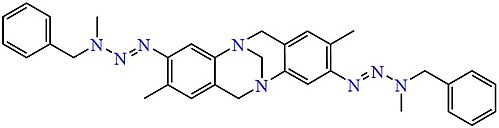
For asymmetrical triazenes, for example (phenyldiazenyl)pyrrolidine (PhN=N−NC4H8), the diazonium salt must be pre-made.Analogues of Tröger's base containing a symmetric pair of asymmetric triazene side-chains have been obtained similarly.[7]
Reactions and applications
Triazenes derived from
Triazenes can be converted to

Triazenes decompose in the presence of protonating or alkylating agents into
Polymeric triazenes are applied as conductive and absorbent materials.[10] Triazenes have been used in the synthesis of cinnoline, functionalized lactams, and coumarins.[9][8]
References
- ^ "Dacarbazine". The American Society of Health-System Pharmacists. Archived from the original on 11 September 2017. Retrieved 8 December 2016.
- ISSN 1433-7851.
- ISBN 978-3527306732.
- ^ .
- ^ S2CID 196805424.
- doi:10.15227/orgsyn.014.0024.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - S2CID 99294625.
- ^ PMID 12298030.
- ^ .
- .