Tuinal
 structure | |
| Combination of | |
|---|---|
| secobarbital | short-acting barbiturate |
| amobarbital | intermediate-acting barbiturate |
| Clinical data | |
| Routes of administration | oral |
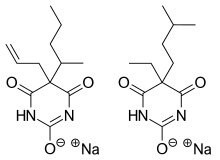
Tuinal was the brand name of a discontinued combination drug composed of two barbiturate salts (secobarbital sodium and amobarbital sodium) in equal proportions.
Tuinal was introduced as a sedative-hypnotic (sleeping pill) medication in the late 1940s by Eli Lilly. It was also used in obstetrics for childbirth.[1][2] It was produced in brightly colored half-reddish orange and half-turquoise blue gelatin capsule form (bullet-shaped Pulvules) for oral administration. Individual capsules contained 50 mg, 100 mg, or 200 mg of barbiturate salts. The combination of a short-acting barbiturate, Secobarbital, with an intermediate-acting barbiturate, Amobarbital, aimed to provide "a rapid yet prolonged hypnotic action".[3]
Eli Lilly has discontinued the manufacture of Tuinal in the United States due to the diminishing use of barbiturates (replaced by the
Abuse

Tuinal saw widespread abuse as a
Tuinal is classified as a Schedule II drug under the Controlled Substances Act in the United States, meaning it requires a prescription from a licensed practitioner.
References
- PMID 20344014.
- JSTOR 25361874.
- JSTOR 3457169.
- ISBN 9781414490144. Retrieved November 4, 2022.
- ISBN 9781414404448. Retrieved November 4, 2022.
- PMID 4301261.
- S2CID 24568454.
