User:FacetiousLobster/sandbox

| |

| |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C32H18N8 | |
| Molar mass | 514.552 g·mol−1 |
| Hazards | |
| GHS labelling: | |
 [1] [1]
| |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Phthalocyanine (H2Pc) is a large,
Properties
Phthalocyanine and derived metal complexes (MPc) tend to aggregate and, thus, have low solubility in common solvents.
There are many derivatives of the parent phthalocyanine, where either carbon atoms of the macrocycle are exchanged for nitrogen atoms or the peripheral hydrogen atoms are substituted by functional groups like
History
In 1907, an unidentified blue compound, now known to be phthalocyanine, was reported.[6] In 1927, Swiss researchers serendipitously discovered copper phthalocyanine, copper naphthalocyanine, and copper octamethylphthalocyanine in an attempted conversion of o-dibromobenzene into phthalonitrile. They remarked on the enormous stability of these complexes but did not further characterize them.[7] In the same year, iron phthalocyanine was discovered at Scottish Dyes of Grangemouth, Scotland (later ICI).[8] It was not until 1934 that Sir Patrick Linstead characterized the chemical and structural properties of iron phthalocyanine.[9]
Synthesis
Phthalocyanine is formed through the cyclotetramerization of various
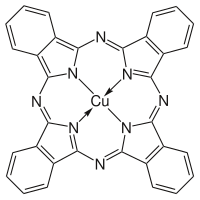
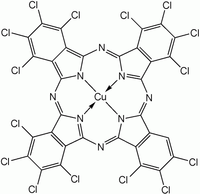
More often, MPc is synthesized rather H2Pc due to the greater research interest in the former. To prepare these complexes, the phthalocyanine synthesis is conducted in the presence of metal salts. Two copper phthalocyanines are shown in the figure below.
.Applications
At the initial discovery of Pc, its uses were primarily limited to dyes and pigments.
Toxicity and hazards
No evidence has been reported for acute toxicity or carcinogenicity of phthalocyanine compounds. The LD50 (rats, oral) is 10 g/kg.[11]
Related compounds

Phthalocyanines are structurally related to other
2Pc.
References
ISBNs for the books listed. . (May 2018) |
- ^ Pubchem. "Pigment blue 16". pubchem.ncbi.nlm.nih.gov. Retrieved 2018-04-08.
- ^ ISSN 0021-9568.
- ISSN 0022-2461.
- .
- ISSN 2040-3372.
- .
- .
- ^ "The Discovery of a New Pigment—The Story of Monastral Blue by Imperial Chemical Industries". colorantshistory.org.
- ISSN 0368-1769.
- doi:10.3390/ma2031127.)
{{cite journal}}: CS1 maint: unflagged free DOI (link - ^ ISBN 978-3527306732..
- PMID 24828002.
- ISSN 0019-7866.
- ISSN 1528-0691.
- ISSN 2046-2069.
- .
- ISSN 0009-2665.
- .
- .
- ISSN 0743-7463.
- .
External links
- Society of Porphyrins and Phthalocyanines
- Sir Patrick Linstead: Phthalocyanines (Video)
- Journal of Porphyrins and Phthalocyanines
- ICI GRANGEMOUTH DISCOVERY VIDEO