Vesicle fusion
Vesicle fusion is the merging of a vesicle with other
Triggers
Stimuli that trigger vesicle fusion act by increasing intracellular Ca2+.
- voltage-dependent calcium channelsthat cause influx of Ca2+ into the cell.
- In the IP3/DAG pathwayto increase Ca2+. Examples of this mechanism include:
Model systems
Model systems consisting of a single
Mechanisms
Synaptic cleft fusion
In synaptic vesicle fusion, the vesicle must be within a few nanometers of the target membrane for the fusion process to begin. This closeness allows the cell membrane and the vesicle to exchange lipids which is mediated by certain proteins which remove water that comes between the forming junction. Once the vesicle is in position it must wait until Ca2+ enters the cell by the propagation of an action potential to the presynaptic membrane.[5] Ca2+ binds to specific proteins, one of which is Synaptotagmin, in neurons which triggers the complete fusion of the vesicle with the target membrane.[6]
SNARE proteins are also thought to help mediate which membrane is the target of which vesicle.[7]
SNARE protein and pore formation
, with membrane lipids and/or the partially assembled SNARE complex.One hypothesis implicates the molecule Complexin within the SNARE complex and its interaction with the molecule synaptotagmin.[9] Known as the "clamp" hypothesis, the presence of complexin normally inhibits the fusion of the vesicle to the cell membrane. However, binding of calcium ions to synaptotagmin triggers the complexin to be released or inactivated, so that the vesicle is then free to fuse.[10]
According to the "zipper" hypothesis, the complex assembly starts at the N-terminal parts of SNARE motifs and proceeds towards the C-termini that anchor interacting proteins in membranes. Formation of the "trans"-SNARE complex proceeds through an intermediate complex composed of SNAP-25 and syntaxin-1, which later accommodates synaptobrevin-2 (the quoted syntaxin and synaptobrevin isotypes participate in neuronal neuromediator release).
Based on the stability of the resultant cis-SNARE complex, it has been postulated that energy released during the assembly process serves as a means for overcoming the repulsive forces between the membranes. There are several models that propose explanation of a subsequent step – the formation of stalk and fusion pore, but the exact nature of these processes remains debated. Two of the most prominent models on fusion pore formation are the lipid-lined and protein-lined fusion pore theories.[11]
Lipid-lined fusion pore theory
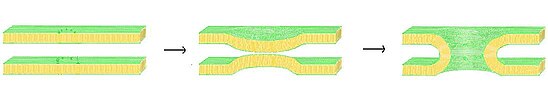
One possible model for fusion pore formation is the lipid-line pore theory. In this model, once the membranes have been brought into sufficiently close proximity via the "zipper" mechanism of the
While a lipid-lined pore is possible and can achieve all the same properties observed in early pore formation, sufficient data does not exist to prove it is the sole method of formation.[13] There is not currently a proposed mechanism on inter-cellular regulation for fluctuation of lipid-lined pores, and they would have a substantially more difficult time producing effects such as the "kiss-and-run" when compared with their protein-lined counterparts. Lipid-lined pores effectiveness would also be highly dependent on the composition of both membranes, and its success or failure could vary wildly with changes in elasticity and rigidity.[13]
Protein-lined fusion pore theory
Another possible model for fusion pore formation is the protein-lined pore theory. In this model, after activation of
A protein-lined pore perfectly meets all the observed requirements of the early fusion pore, and while some data does support this theory,[14] sufficient data does not exist to pronounce it the primary method of fusion. A protein-lined pore requires at least five copies of the SNARE complex while fusion has been observed with as few as two.[14]
In both theories the function of the SNARE complex remains largely unchanged, and the entire SNARE complex is necessary to initiate fusion. It has, however, been proven that in vitro Syntaxin per se is sufficient to drive spontaneous calcium independent fusion of synaptic vesicles containing v-SNAREs.[15] This suggests that in Ca2+-dependent neuronal exocytosis synaptotagmin is a dual regulator, in absence of Ca2+ ions to inhibit SNARE dynamics, while in presence of Ca2+ ions to act as agonist in the membrane fusion process.
Kiss-and-run hypothesis
In
There is some indication that vesicles may only form a small pore in the presynaptic membrane allowing contents to be released by standard diffusion for a short while before retreating back into the presynaptic cell. This mechanism may be a way around
"Kiss and run" has been shown to occur in endocrine cells, though it has not been directly witnessed in synaptic gaps.[16]
See also
- SNARE
- Presynaptic active zone
- membrane fusionstudies.
References
- ^ ISBN 978-0-7817-7311-9.
- ISBN 978-1-4160-2328-9.
- S2CID 1465571.
- ^ sciencedirect
- ISBN 978-0-12-088397-4.
- ^ Pigino et al. p 158
- ^ Pigino et al. p.143
- S2CID 199021983.
- ^ PMID 21785414.
- ^ Richmond, Janet. "Synapse Function".
- PMID 16689631.
- PMID 16220905.
- ^ PMID 1420930.
- ^ PMID 25855187.
- S2CID 37732173. Archived from the original(PDF) on 2011-07-19. Retrieved 2009-05-31.
- ^ Piginio et al. pp. 161-162

