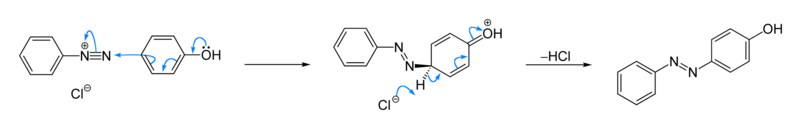
Azo coupling
In
Uses of the reaction
Aromatic azo compounds tend to be brightly colored due to their extended
Azo printing exploits this reaction as well. In this case, the diazonium ion is degraded by light, leaving a latent image in undegraded diazonium salt which is made to react with a phenol, producing a colored image: the blueprint.[3]
The reaction is also used in the Pauly reaction test to detect tyrosine or histidine residues in proteins.
Additionally, through the azo coupling reaction between the aromatic diazonium ion and aromatic amino acid residues, this reaction also be used to form or to modify proteins such as tRNA synthetase.[4]
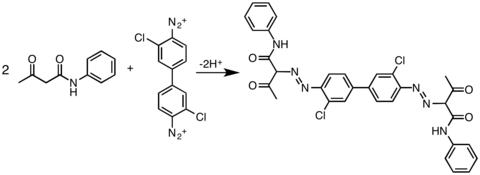
Examples of azo C-coupling reactions
Illustrative is the reaction of
Naphthols are popular coupling agents. One example is the synthesis of the dye "organol brown" from aniline and 1-naphthol:
Similarly,
Besides activated aromatic coupling agents, other nucleophilic carbons could also be coupled with diazonium salt:
Examples of azo N-coupling reactions

In alkaline media, diazonium salt can react with most primary and secondary amines, which exist as a free base to produce triazene.[8] This chemical reaction is called azo N-coupling,[9] or the synthesis of azoamines.[10]
The dye called aniline yellow is produced by the reaction of aniline and a diazonium salt. In this case the C- and N-coupling compete.[2]
References
- ISBN 978-0-471-72091-1
- ^ ISBN 978-3527306732.
- ISBN 978-3527306732.
- PMID 28787141.
- .
- .
- .
- S2CID 196805424.
- .
- ^ Serge Ratton, Bernard Botannet (1981). "Preparation of aromatic azoamines by diazotization/coupling/rearrangement of aromatic amines". US Patent 4275003A.