Electrophile
In
, have an atom that carries a partial positive charge, or have an atom that does not have an octet of electrons.Electrophiles mainly interact with nucleophiles through
Organic chemistry
Addition of halogens
These occur between alkenes and electrophiles, often halogens as in
- C2H4 + Br2 → BrCH2CH2Br
This takes the form of 3 main steps shown below;[3]
- Forming of a π-complex
- The electrophilic Br-Br molecule interacts with electron-rich alkene molecule to form a π-complex1.
- The electrophilic Br-Br molecule interacts with electron-rich alkene molecule to form a
- Forming of a three-membered bromonium ion
- The alkene is working as an electron donor and bromine as an electrophile. The three-membered bromonium ion2 consisted of two carbon atoms and a bromine atom forms with a release of Br−.
- The alkene is working as an electron donor and bromine as an electrophile. The three-membered
- Attacking of bromide ion
- The bromonium ion is opened by the attack of Br− from the back side. This yields the antiperiplanarconfiguration. When other nucleophiles such as water or alcohol are existing, these may attack 2 to give an alcohol or an ether.
- The bromonium ion is opened by the attack of Br− from the back side. This yields the
This process is called AdE2 mechanism ("addition, electrophilic, second-order").
Addition of hydrogen halides
Hydrogen halides such as hydrogen chloride (HCl) adds to alkenes to give alkyl halides in hydrohalogenation. For example, the reaction of HCl with ethylene furnishes chloroethane. The reaction proceeds with a cation intermediate, being different from the above halogen addition. An example is shown below:
- Proton (H+) adds (by working as an electrophile) to one of the carbon atoms on the alkene to form cation 1.
- Chloride ion (Cl−) combines with the cation 1 to form the adducts 2 and 3.
In this manner, the stereoselectivity of the product, that is, from which side Cl− will attack relies on the types of alkenes applied and conditions of the reaction. At least, which of the two carbon atoms will be attacked by H+ is usually decided by Markovnikov's rule. Thus, H+ attacks the carbon atom that carries fewer substituents so as the more stabilized carbocation (with the more stabilizing substituents) will form.
This is another example of an AdE2 mechanism.[5] Hydrogen fluoride (HF) and hydrogen iodide (HI) react with alkenes in a similar manner, and Markovnikov-type products will be given. Hydrogen bromide (HBr) also takes this pathway, but sometimes a radical process competes and a mixture of isomers may form. Although introductory textbooks seldom mentions this alternative,[6] the AdE2 mechanism is generally competitive with the AdE3 mechanism (described in more detail for alkynes, below), in which transfer of the proton and nucleophilic addition occur in a concerted manner. The extent to which each pathway contributes depends on the several factors like the nature of the solvent (e.g., polarity), nucleophilicity of the halide ion, stability of the carbocation, and steric effects. As brief examples, the formation of a sterically unencumbered, stabilized carbocation favors the AdE2 pathway, while a more nucleophilic bromide ion favors the AdE3 pathway to a greater extent compared to reactions involving the chloride ion.[7]
In the case of dialkyl-substituted alkynes (e.g., 3-hexyne), the intermediate vinyl cation that would result from this process is highly unstable. In such cases, the simultaneous protonation (by HCl) and attack of the alkyne by the nucleophile (Cl−) is believed to take place. This mechanistic pathway is known by the Ingold label AdE3 ("addition, electrophilic, third-order"). Because the simultaneous collision of three chemical species in a reactive orientation is improbable, the termolecular transition state is believed to be reached when the nucleophile attacks a reversibly-formed weak association of the alkyne and HCl. Such a mechanism is consistent with the predominantly anti addition (>15:1 anti:syn for the example shown) of the hydrochlorination product and the termolecular rate law, Rate = k[alkyne][HCl]2.[8][9] In support of the proposed alkyne-HCl association, a T-shaped complex of an alkyne and HCl has been characterized crystallographically.[10]
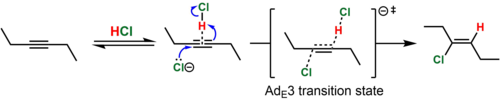
In contrast, phenylpropyne reacts by the AdE2ip ("addition, electrophilic, second-order, ion pair") mechanism to give predominantly the syn product (~10:1 syn:anti). In this case, the intermediate vinyl cation is formed by addition of HCl because it is resonance-stabilized by the phenyl group. Nevertheless, the lifetime of this high energy species is short, and the resulting vinyl cation-chloride anion ion pair immediately collapses, before the chloride ion has a chance to leave the solvent shell, to give the vinyl chloride. The proximity of the anion to the side of the vinyl cation where the proton was added is used to rationalize the observed predominance of syn addition.[7]

Hydration
One of the more complex
- C2H4 + H2O → C2H5OH
As can be seen, the H2SO4 does take part in the overall reaction, however it remains unchanged so is classified as a catalyst.
This is the reaction in more detail:
- The H–OSO3H molecule has a δ+ charge on the initial H atom. This is attracted to and reacts with the double bond in the same way as before.
- The remaining (negatively charged) −OSO3H ion then attaches to the carbocation, forming ethyl hydrogensulphate (upper way on the above scheme).
- When water (H2O) is added and the mixture heated, ethanol (C2H5OH) is produced. The "spare" hydrogen atom from the water goes into "replacing" the "lost" hydrogen and, thus, reproduces sulfuric acid. Another pathway in which water molecule combines directly to the intermediate carbocation (lower way) is also possible. This pathway become predominant when aqueous sulfuric acid is used.
Overall, this process adds a molecule of water to a molecule of ethene.
This is an important reaction in industry, as it produces ethanol, whose purposes include fuels and starting material for other chemicals.
Chiral derivatives
Many electrophiles are chiral and optically stable. Typically chiral electrophiles are also optically pure.
One such

Polymer-bound chiral
Electrophilicity scale
| Fluorine | 3.86 |
| Chlorine | 3.67 |
| Bromine | 3.40 |
| Iodine | 3.09 |
| Hypochlorite | 2.52 |
| Sulfur dioxide | 2.01 |
| Carbon disulfide | 1.64 |
| Benzene | 1.45 |
| Sodium | 0.88 |
| Some selected values [14] (no dimensions) | |
Several methods exist to rank electrophiles in order of reactivity[15] and one of them is devised by Robert Parr[14] with the electrophilicity index ω given as:
with the electronegativity and
where is the
An electrophilicity index also exists for
Superelectrophiles
Superelectrophiles are defined as cationic electrophilic reagents with greatly enhanced reactivities in the presence of
2BF−
4 only in presence of a strong acid like fluorosulfuric acid
In gitionic (gitonic) superelectrophiles, charged centers are separated by no more than one atom, for example, the protonitronium ion O=N+=O+—H (a protonated nitronium ion). And, in distonic superelectrophiles, they are separated by 2 or more atoms, for example, in the fluorination reagent F-TEDA-BF4.[18]
See also
- Nucleophile
- TRPA1,[19][20] the sensory neural target for electrophilic irritants in mammals.
References
- ^ "Nucleophiles and Electrophiles". butane.chem.uiuc.edu. Archived from the original on 2020-08-01. Retrieved 2020-09-21.
- ^ "Electrophile | chemistry". Encyclopedia Britannica. Retrieved 2020-09-21.
- PMID 12596140.
- .
- ^ In analogy to aromatic substitution, this process has also been termed an A-SE2 mechanism.
- )
- ^ )
- ISSN 0002-7863.
- )
- ISSN 0002-7863.
- .
- .
- PMID 11529793.
- ^ .
- PMID 16771443.
- PMID 17559221.
- .
- PMID 17999519.
- PMID 31447178.
- PMID 32641835.









