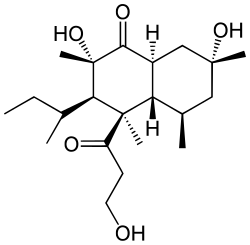
Betaenone B
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
JSmol) | |
| Melting point | 103.5 to 108 °C (218.3 to 226.4 °F) |
| |
| |
| (verify) | |
Betaenone B, like other
protein synthesis.[5] Most of the major work on betaenone B, including the initial structure elucidation of betaenone A, B and C as well as the partial elucidation mechanism of biosynthesis, was presented in three short papers published between 1983 and 1988.[1][2][3] The compounds were found to inhibit a variety of protein kinases signifying a possible role in cancer treatment.[6]
The structure of betaenone B was determined via
Bioactivity
of this synthetic product was not tested and no further work on total synthesis of betaenones has been published since.
While a complete de novo synthesis of betaenone B has yet to be reported, Daniel Pratt and Paul Hopkins in 1988 proposed a method for synthesizing a precursor of betaenone B from methoxybenzoquinone and 1,3-butadiene via a Diels–Alder reaction and Claisen chemistry[clarification needed].[7]
Biosynthesis
Very little work has been done to elucidate the biosynthetic pathways of betaenones with almost no literature references published on the subject since 1988. Their low phytotoxicity and lack of biological significance in
cytochrome P-450
is responsible for the oxidation at these three positions since its inhibition produces probetaenone 1, the non-oxidized form of betaenone B.
References
- ^ .
- ^ .
- ^ .
- ^ Afonin AN (2008). "Interactive Agricultural Ecological Atlas of Russia and Neighboring Countries. Economic Plants and their Diseases, Pests and Weeds Online".
- ^ PMID 6402759.
- .
- ^ .


