CAMSAP2
| CAMSAP2 | |||
|---|---|---|---|
| Identifiers | |||
Gene ontology | |||
| Molecular function | |||
| Cellular component | |||
| Biological process |
| ||
| Sources:Amigo / QuickGO | |||
Ensembl | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) | |||||||||
| RefSeq (protein) | |||||||||
| Location (UCSC) | Chr 1: 200.74 – 200.86 Mb | Chr 1: 136.2 – 136.27 Mb | |||||||
| PubMed search | [3] | [4] | |||||||
| View/Edit Human | View/Edit Mouse |
Calmodulin-regulated spectrin-associated protein family member 2 (CAMSAP2) is a protein that, in humans, is encoded by the CAMSAP2 gene.[5] CAMSAP2 possesses a microtubule-binding domain near the C-terminal region where said microtubule interactions occur. On these C-terminal regions, protein-protein interactions are accelerated by three coiled-coil domains, which function as molecular spacers.[6] CAMSAP2 acts as a microtubule minus-end anchor and binds microtubules through its CKK domain. CAMSAP2 is necessary for the proper organization and stabilization of interphase microtubules. The protein also plays a vital role in cell migration.[7] CAMSAP2 can stabilize and attach microtubule minus ends to the Golgi through the AKAP9 complex and myomegalin. CLASP1 proteins responsible for microtubule stability are not required for the Golgi tethering. When no centromeres are present, AKAP9 and CAMSAP-2 dependent pathways of these microtubule minus ends become a dominant force and must exist in order to observe the maintenance of microtubule density.[8]
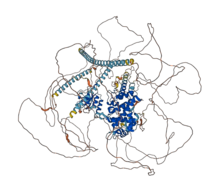
Structure
Microtubules are cytoskeletal polymers with structurally and functionally different ends. There exists a plus-end and a minus-end on each microtubule. The CAMSAP family of proteins contributes to regulating the minus-ends of microtubules.[12] CAMSAP2 contains a CKK domain that binds to microtubules. This CKK domain is a defining factor of the CAMSAP protein family. It enables CAMSAP2 to recognize and bind to microtubule minus ends and allows CAMSAP2 to decorate and stabilize the microtubule lattice formed by minus-end polymerization.[13] In addition to a CKK domain, CAMSAP2 contains an N-terminal calponin homology domain involved with actin binding.[14]
Function
Non-centrosomal microtubule maintenance
CAMSAP2 proteins are observed to be primarily responsible for the maintenance of non-centrosomal microtubules. In epithelial cells, major microtubules are not anchored to the centrosome, which can be observed in other cell types. CAMSAP2 cooperates with calmodulin-regulated spectrin-associated protein 3 (CAMSAP3) to achieve the organization observed in these non-centrosomal microtubules. They possess the ability to suppress the organization of microtubules by the centrosome, and this family of proteins is important for the suitable arrangement of organelles in the cell body. These proteins gathered at the minus ends of these non-centrosomal microtubules can stabilize them. Without these proteins, the Golgi apparatus would exhibit irregular distributions of the microtubules.[15]
Pancreatic islet cells
In pancreatic β-cells, glucose stimulation leads to the remodeling of microtubules responsible for insulin secretion. CAMSAP2 binds to the minus ends of microtubules in normal clonal cells. The knockdown of CAMSAP2 in these β-cells reduces the total insulin content secreted through glucose-stimulated insulin secretion. However, CAMSAP2 localizes to the Golgi apparatus instead of the microtubule minus ends. This oddity is observed only in β-cells as opposed to α-cells. With this collection at the Golgi apparatus, CAMSAP2 promotes the protein trafficking of the Golgi, efficiently facilitating the process. Without CAMSAP2, there would not be adequate insulin production for secretion from these β-cells.[16]
Migration
CAMSAP2-dependent microtubule organization promotes directional
Microtubule nucleation

The initial polymerization of microtubules can be called
Regulation
Regulator of neuronal polarity and development
CAMSAP2 is responsible for controlling
Regulator of blood–testis barrier (BTB)
CAMSAP2 is involved in targeting microtubule minus-ends in
Clinical research
Hepatocellular carcinoma (HCC)
CAMSAP2 plays a significant role in the migration of cancer cells. It has been observed that CAMSAP2 is severely upregulated in cancers such as
Colorectal cancer
CAMPSAP promotes the migration of colorectal cancer cells by activating the
Gastric cancer
An association between CAMSAP2 expression levels and the progression and prognosis of gastric cancer was investigated in 2023.
See also
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000118200 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000041570 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "CAMSAP2 calmodulin regulated spectrin associated protein family member 2 [Homo sapiens (human)] - Gene - NCBI". www.ncbi.nlm.nih.gov. Archived from the original on 2024-02-27. Retrieved 2024-03-11.
- from the original on 2024-04-15. Retrieved 2024-04-15.
- from the original on 2022-06-15. Retrieved 2022-09-29.
- from the original on 2024-04-15. Retrieved 2024-04-15.
- ^ "AlphaFold Protein Structure Database". alphafold.ebi.ac.uk. Archived from the original on 2022-10-01. Retrieved 2024-04-12.
- from the original on 2024-04-15. Retrieved 2024-04-15.
- PMID 34265844.
- PMID 31748546.
- ^ "UniProt". www.uniprot.org. Archived from the original on 2024-04-15. Retrieved 2024-04-14.
- from the original on 2023-02-24. Retrieved 2024-04-15.
- PMID 23169647.
- from the original on 2024-04-15. Retrieved 2024-04-15.
- from the original on 2024-04-15. Retrieved 2024-04-15.
- PMID 35762204.
- PMID 35762204.
- from the original on April 15, 2024. Retrieved April 15, 2024.
- ^ PMID 30994903.
- from the original on 2024-03-11. Retrieved 2024-04-15.
- PMID 36207462.
- ^ PMID 37814859.
- PMID 21295093.
Further reading
- Baines AJ, Bignone PA, King MD, Maggs AM, Bennett PM, Pinder JC, et al. (September 2009). "The CKK domain (DUF1781) binds microtubules and defines the CAMSAP/ssp4 family of animal proteins". Molecular Biology and Evolution. 26 (9): 2005–2014. PMID 19508979.
