Chan–Lam coupling
| Chan-Lam coupling | |
|---|---|
| Named after | Dominic Chan Patrick Lam |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | chan-lam-coupling |
| RSC ontology ID | RXNO:0000374 |
The Chan–Lam coupling reaction – also known as the Chan–Evans–Lam coupling is a
History
Dominic Chan, David Evans, and Patrick Lam published their work nearly simultaneously.[2][3][4][5][6] The mechanism however remained uncertain for many years. Later developments by others extended the scope to include using carboxylic acids, giving aryl-ester products.[7]
Mechanism
Analysis of the mechanism is complicated by the lability of copper reagents and the multicomponent nature of the reaction.[8] The reaction proceeds via the formation of copper-aryl complexes. A copper(III)-aryl-alkoxide or copper(III)-aryl-amide intermediate undergoes Reductive elimination to give the aryl ether or aryl amine, respectively:
- Ar-Cu(III)-NHR-L2 → Ar-NHR + Cu(I)L2
- Ar-Cu(III)-OR-L2 → Ar-OR + Cu(I)L2
Example
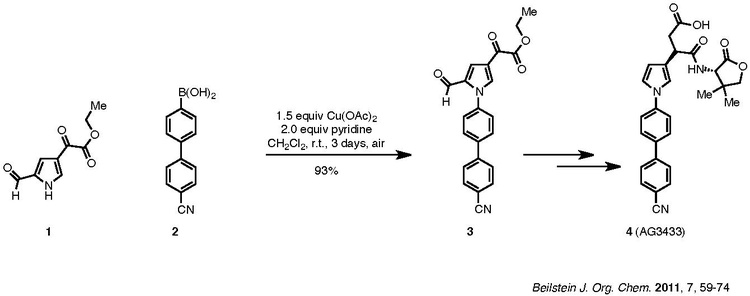
An example of the Chan–Lam coupling to synthesize biologically active compounds is shown below:
Compound 1, a pyrrole, is coupled with aryl boronic acid, 2, to afford product 3, which is then carried forward to the target 4. The
Further reading
- Kodepelly Sanjeeva Rao; Tian-Shung Wu (2012). "Chan-Lam coupling reactions: synthesis of heterocycles". Tetrahedron. 68 (38): 7735–7754. .
References
- ISBN 9783527639328.
- .
- .
- .
- .
- .
- PMID 20942492.
- ^
Vantourout, J. C.; Miras, H. N.; Isidro-Llobet, A.; Sproules, S.; Watson, A. J. B. (2017). "Spectroscopic Studies of the Chan–Lam Amination: A Mechanism-Inspired Solution to Boronic Ester Reactivity" (PDF). Journal of the American Chemical Society. 139 (13): 4769–4779. PMID 28266843.