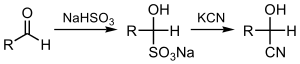
Cyanohydrin
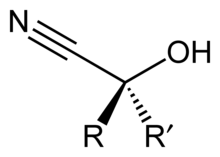
In ,
- RR’C=O + HCN → RR’C(OH)CN
In this reaction, the
Cyanohydrins are intermediates in the
Acetone cyanohydrins
Preparative methods
Cyanohydrins were first prepared by the addition of HCN and a catalyst (base or enzyme) to the corresponding carbonyl.[5] On a laboratory scale the use of HCN (toxic) is largely not encouraged, for this reason other less dangerous cyanation reagents are sought out. In situ formation of HCN can be sourced using precursors such as acetone cyanohydrin. Alternatively, cyano-silyl derivatives such as TMS-CN allows for both the cyanation and protection in one step without the need for HCN.[6] Similar procedures relying on ester, phosphate and carbonate formation have been reported.[7][8][9]

Other cyanohydrins

Glycolonitrile, also called hydroxyacetonitrile or formaldehyde cyanohydrin, is the organic compound with the formula HOCH2CN. It is the simplest cyanohydrin, being derived from formaldehyde.[10]
-
glycolonitrile
-
acetone cyanohydrin
-
mandelonitrile
See also
References
- ^ PMID 18914000.
- ^ a b Corson, B. B.; Dodge, R. A.; Harris, S. A.; Yeaw, J. S. (1941). "Mandelic Acid". Organic Syntheses; Collected Volumes, vol. 1, p. 336.
- . Article Online Posting Date: June 15, 2000
- ^ North, M.; Usanov, D. L.; Young, C. Lewis Acid Catalyzed Asymmetric Cyanohydrin Synthesis. Chem. Rev. 2008, 108 (12), 5146–5226. doi:10.1021/cr800255k.
- ^ Lidy, W.; Sundermeyer, W. Spaltungsreaktionen Des Trimethylsilylcyanids, Eine Neue Darstellungsmethode Für O-(Trimethylsilyl)Cyanhydrine. Chem. Ber. 1973, 106 (2), 587–593. doi:10.1002/cber.19731060224
- ^ Scholl, M.; Lim, C. K.; Fu, G. C. Convenient and Efficient Conversion of Aldehydes to Acylated Cyanohydrins Using Tributyltin Cyanide as a Catalyst. J. Org. Chem. 1995, 60 (19), 6229–6231. doi: 10.1021/jo00124a052.
- ^ Yoneda, R.; Harusawa, S.; Kurihara, T. Cyano Phosphate: An Efficient Intermediate for the Chemoselective Conversion of Carbonyl Compounds to Nitriles. J. Org. Chem. 1991, 56 (5), 1827–1832. doi: 10.1021/jo00005a031.
- ^ Juhl, M.; Petersen, A. R.; Lee, J.-W. CO2‐Enabled Cyanohydrin Synthesis and Facile Iterative Homologation Reactions. Chem. – A Eur. J. 2020. doi: 10.1002/chem.202003623.
- ^ Gaudry, R. (1955). "Glycolonitrile". Organic Syntheses; Collected Volumes, vol. 3, p. 436.
External links
- Gold Book definition of cyanohydrins [1]