Hydrogen cyanide
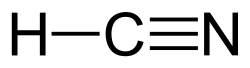 | |||
| |||
 | |||
| Names | |||
|---|---|---|---|
| IUPAC name
Formonitrile[2]
| |||
| Systematic IUPAC name
Methanenitrile[2] | |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.000.747 | ||
| EC Number |
| ||
| KEGG | |||
| MeSH | Hydrogen+Cyanide | ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1051 | ||
CompTox Dashboard (EPA)
|
|||
| |||
SMILES
| |||
| Properties | |||
| HCN | |||
| Molar mass | 27.0253 g/mol | ||
| Appearance | Colorless liquid or gas | ||
| Odor | bitter almond-like[3] | ||
| Density | 0.6876 g/cm3[4] | ||
| Melting point | −13.29 °C (8.08 °F; 259.86 K)[4] | ||
| Boiling point | 26 °C (79 °F; 299 K)[4]: 4.67 | ||
| Miscible | |||
| Solubility in ethanol | Miscible | ||
| Vapor pressure | 100 kPa (25 °C)[4]: 6.94 | ||
Henry's law
constant (kH) |
75 μmol Pa−1 kg−1 | ||
| Acidity (pKa) | 9.21 (in water),
12.9 (in DMSO)[5] | ||
| Basicity (pKb) | 4.79 (cyanide anion) | ||
Conjugate acid
|
Hydrocyanonium
| ||
Conjugate base
|
Cyanide | ||
Refractive index (nD)
|
1.2675[6] | ||
| Viscosity | 0.183 mPa·s (25 °C)[4]: 6.231 | ||
| Structure | |||
| tetragonal (>170 K) orthorhombic (<170 K)[7] | |||
| C∞v | |||
| Linear | |||
| 2.98 D | |||
| Thermochemistry | |||
Heat capacity (C)
|
35.9 J K−1 mol−1 (gas)[4]: 5.19 | ||
Std molar
entropy (S⦵298) |
201.8 J K−1 mol−1 | ||
Std enthalpy of (ΔfH⦵298)formation |
135.1 kJ mol−1 | ||
| Hazards | |||
| GHS labelling: | |||
   
| |||
| Danger | |||
| H225, H300+H310+H330, H319, H336, H370, H410 | |||
| P210, P261, P305+P351+P338 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | −17.8 °C (0.0 °F; 255.3 K) | ||
| 538 °C (1,000 °F; 811 K) | |||
Explosive limits
|
5.6% – 40.0%[8] | ||
| Lethal dose or concentration (LD, LC): | |||
LC50 (median concentration)
|
501 ppm (rat, 5 min) 323 ppm (mouse, 5 min) 275 ppm (rat, 15 min) 170 ppm (rat, 30 min) 160 ppm (rat, 30 min) 323 ppm (rat, 5 min)[9] | ||
LCLo (lowest published)
|
200 ppm (mammal, 5 min) 36 ppm (mammal, 2 hr) 107 ppm (human, 10 min) 759 ppm (rabbit, 1 min) 759 ppm (cat, 1 min) 357 ppm (human, 2 min) 179 ppm (human, 1 hr)[9] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 10 ppm (11 mg/m3) [skin][8] | ||
REL (Recommended)
|
ST 4.7 ppm (5 mg/m3) [skin][8] | ||
IDLH (Immediate danger) |
50 ppm[8] | ||
| Related compounds | |||
Related alkanenitriles
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hydrogen cyanide (formerly known as prussic acid) is a
.Whether hydrogen cyanide is an
Structure and general properties
Hydrogen cyanide is a linear molecule, with a triple bond between carbon and nitrogen. The tautomer of HCN is HNC, hydrogen isocyanide.[citation needed]
Smell
Much literature has historically claimed that hydrogen cyanide smells of
About half of people are unable to
The volatile compound has been used as inhalation rodenticide and human poison, as well as for killing whales.[15] Cyanide ions interfere with iron-containing respiratory enzymes.[citation needed]
Chemical properties
Hydrogen cyanide is weakly
Hydrogen cyanide reacts with alkenes to give nitriles. The conversion, which is called hydrocyanation, employs nickel complexes as catalysts.[17]
- RCH=CH2 + HCN → RCH2−CH2CN
Four molecules of HCN will tetramerize into diaminomaleonitrile.[18]
- HgO + 2 HCN → Hg(CN)2 + H2O
History of discovery and naming
Hydrogen cyanide was first isolated in 1752 by French chemist Pierre Macquer who converted Prussian blue to an iron oxide plus a volatile component and found that these could be used to reconstitute it.[20] The new component was what is now known as hydrogen cyanide. It was subsequently prepared from Prussian blue by the Swedish chemist Carl Wilhelm Scheele in 1782,[21] and was eventually given the German name Blausäure (lit. "Blue acid") because of its acidic nature in water and its derivation from Prussian blue. In English, it became known popularly as prussic acid.
In 1787, the French chemist Claude Louis Berthollet showed that prussic acid did not contain oxygen,[22] an important contribution to acid theory, which had hitherto postulated that acids must contain oxygen[23] (hence the name of oxygen itself, which is derived from Greek elements that mean "acid-former" and are likewise calqued into German as Sauerstoff).
In 1811, Joseph Louis Gay-Lussac prepared pure, liquified hydrogen cyanide,[24] and in 1815 he deduced Prussic acid's chemical formula.[25]
Etymology
The word cyanide for the radical in hydrogen cyanide was derived from its French equivalent, cyanure, which Gay-Lussac constructed from the Ancient Greek word κύανος for dark blue enamel or lapis lazuli, again owing to the chemical’s derivation from Prussian blue. Incidentally, the Greek word is also the root of the English color name cyan.
Production and synthesis
The most important process is the
- 2 CH4 + 2 NH3 + 3 O2 → 2 HCN + 6 H2O
In 2006, between 500 million and 1 billion pounds (between 230,000 and 450,000 t) were produced in the US.[27] Hydrogen cyanide is produced in large quantities by several processes and is a recovered waste product from the manufacture of acrylonitrile.[10]
Of lesser importance is the
- CH4 + NH3 → HCN + 3H2
This reaction is akin to steam reforming, the reaction of methane and water to give carbon monoxide and hydrogen.
In the Shawinigan Process,
In the laboratory, small amounts of HCN are produced by the addition of acids to cyanide salts of
- H+ + CN− → HCN
This reaction is sometimes the basis of accidental poisonings because the acid converts a nonvolatile cyanide salt into the gaseous HCN.
Hydrogen cyanide could be obtained from potassium ferricyanide and acid:
Historical methods of production
The large demand for cyanides for mining operations in the 1890s was met by
Applications
HCN is the precursor to
HCN is used globally as a
Occurrence
HCN is obtainable from

On Titan
HCN has been measured in Titan's atmosphere by four instruments on the Cassini space probe, one instrument on Voyager, and one instrument on Earth.[40] One of these measurements was in situ, where the Cassini spacecraft dipped between 1,000 and 1,100 km (620 and 680 mi) above Titan's surface to collect atmospheric gas for mass spectrometry analysis.[41] HCN initially forms in Titan's atmosphere through the reaction of photochemically produced methane and nitrogen radicals which proceed through the H2CN intermediate, e.g., (CH3 + N → H2CN + H → HCN + H2).[42][43] Ultraviolet radiation breaks HCN up into CN + H; however, CN is efficiently recycled back into HCN via the reaction CN + CH4 → HCN + CH3.[42]
On the young Earth
It has been postulated that carbon from a cascade of asteroids (known as the Late Heavy Bombardment), resulting from interaction of Jupiter and Saturn, blasted the surface of young Earth and reacted with nitrogen in Earth's atmosphere to form HCN.[44]
In mammals
Some authors[
It has also been shown that, while stimulating
The
HCN is a constituent of tobacco smoke.[48]
HCN and the origin of life
Hydrogen cyanide has been discussed as a precursor to amino acids and nucleic acids, and is proposed to have played a part in the
In space
HCN has been detected in the interstellar medium[52] and in the atmospheres of carbon stars.[53] Since then, extensive studies have probed formation and destruction pathways of HCN in various environments and examined its use as a tracer for a variety of astronomical species and processes. HCN can be observed from ground-based telescopes through a number of atmospheric windows.[54] The J=1→0, J=3→2, J= 4→3, and J=10→9 pure rotational transitions have all been observed.[52][55][56]
HCN is formed in
HCN is destroyed in interstellar clouds through a number of mechanisms depending on the location in the cloud.
On 11 August 2014, astronomers released studies, using the
In February 2016, it was announced that traces of hydrogen cyanide were found in the atmosphere of the hot Super-Earth 55 Cancri e with NASA's Hubble Space Telescope.[64]
On 14 December 2023, astronomers reported the first time discovery, in the
As a poison and chemical weapon
In World War I, hydrogen cyanide was used by the French from 1916 as a chemical weapon against the Central Powers, and by the United States and Italy in 1918. It was not found to be effective enough due to weather conditions.[68][69] The gas is lighter than air and rapidly disperses up into the atmosphere. Rapid dilution made its use in the field impractical. In contrast, denser agents such as phosgene or chlorine tended to remain at ground level and sank into the trenches of the Western Front's battlefields. Compared to such agents, hydrogen cyanide had to be present in higher concentrations in order to be fatal.
A hydrogen cyanide concentration of 100–200
The Chemical Weapons Convention lists it under Schedule 3 as a potential weapon which has large-scale industrial uses. Signatory countries must declare manufacturing plants that produce more than 30 metric tons per year, and allow inspection by the Organisation for the Prohibition of Chemical Weapons.
Perhaps its most infamous use is
During
Hydrogen cyanide was also the agent employed in judicial execution in some U.S. states, where it was produced during the execution by the action of sulfuric acid on sodium cyanide or potassium cyanide.[76]
Under the name prussic acid, HCN has been used as a killing agent in whaling harpoons, although it proved quite dangerous to the crew deploying it, and it was quickly abandoned.[15] From the middle of the 18th century it was used in a number of poisoning murders and suicides.[77]
Hydrogen cyanide gas in air is explosive at concentrations above 5.6%.[78]
References
- ^ "hydrogen cyanide (CHEBI:18407)". Chemical Entities of Biological Interest. UK: European Bioinformatics Institute. 18 October 2009. Main. Retrieved 2012-06-04.
- ^ a b c "Hydrogen Cyanide". PubChem. National Center for Biotechnology Information.
- ISSN 1020-6167.
- ^ ISBN 978-1439855119.
- ^ Evans DA. "pKa's of Inorganic and Oxo-Acids" (PDF). Archived (PDF) from the original on 2022-10-09. Retrieved June 19, 2020.
- ISBN 978-0070494398.
- ISSN 0193-4929.
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0333". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "Hydrogen cyanide". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ ISBN 978-3-527-30673-2.
- ^ "Human Metabolome Database: Showing metabocard for Hydrogen cyanide (HMDB0060292)".
- ^ "How do people know HCN smells like almonds?".
- ^ "Do almonds smell like they do because of cyanide?".
- ^ "Cyanide, inability to smell". Online Mendelian Inheritance in Man. Retrieved 2010-03-31.
- ^ a b Lytle T. "Poison Harpoons". Whalecraft.net. Archived from the original on 2019-02-15.
- PMID 32027458.
- OCLC 54966334.
- .
- ^ F. Wagenknecht; R. Juza (1963). "Mercury (II) cyanide". In G. Brauer (ed.). Handbook of Preparative Inorganic Chemistry. Vol. 2 (2nd ed.). NY, NY: Academic Press.
- ^ Macquer PJ (1756). "Éxamen chymique de bleu de Prusse" [Chemical examination of Prussian blue]. Mémoires de l'Académie royale des Sciences (in French): 60–77.
- ^ Scheele CW (1782). "Försök, beträffande det färgande ämnet uti Berlinerblå" [Experiment concerning the coloring substance in Berlin blue]. Kungliga Svenska Vetenskapsakademiens Handlingar (Royal Swedish Academy of Science's Proceedings (in Swedish). 3: 264–275.
Reprinted in Latin as: Scheele CW, Hebenstreit EB, eds. (1789). "De materia tingente caerulei berolinensis". Opuscula Chemica et Physica [The dark matter tingente caerulei berolinensis] (in Latin). Vol. 2. Translated by Schäfer GH. (Leipzig ("Lipsiae") (Germany): Johann Godfried Müller. pp. 148–174. - ^ Berthollet CL (1789). "Mémoire sur l'acide prussique" [Memoir on prussic acid]. Mémoires de l'Académie Royale des Sciences (in French): 148–161.
Reprinted in: Berthollet CL (1789). "Extrait d'un mémoire sur l'acide prussique" [Extract of a memoir on prussic acid]. Annales de Chimie. 1: 30–39. - ^ Newbold BT (1999-11-01). "Claude Louis Berthollet: A Great Chemist in the French Tradition". Canadian Chemical News. Archived from the original on 2008-04-20. Retrieved 2010-03-31.
- ^ Gay-Lussac JL (1811). "Note sur l'acide prussique" [Note on prussic acid]. Annales de Chimie. 44: 128–133.
- ^ Gay-Lussac JL (1815). "Recherche sur l'acide prussique" [Research on prussic acid]. Annales de Chimie. 95: 136–231.
- .
- ^ "Non-confidential 2006 IUR Records by Chemical, including Manufacturing, Processing and Use Information". EPA. Archived from the original on 2013-05-10. Retrieved 2013-01-31.
- .
- ^ "MSDS for potassium ferricyanide" (PDF). Archived from the original (PDF) on 2016-04-18. Retrieved 2023-04-17.
- ^ "Potassium ferricyanide". PubChem. National Center for Biotechnology Information.
- ^ "Manual of fumigation for insect control – Space fumigation at atmospheric pressure (Cont.)". Food and Agriculture Organization.
- ^ "New greenhouse gas identified". News.mit.edu. 11 March 2009.
- ^ "Chapter 10 : Methyl Bromide" (PDF). Csl.noaa.gov. Archived (PDF) from the original on 2022-10-09.
- PMID 10669009.
- PMID 9431670.
- ^ "Are Apple Cores Poisonous?". The Naked Scientists. 26 September 2010. Archived from the original on 6 March 2014. Retrieved 6 March 2014.
- PMID 1650055.
- S2CID 40193390.
- PMID 29751568.
- .
- .
- ^ S2CID 221095540.
- S2CID 73442008.
- ^ Wade N (2015-05-04). "Making Sense of the Chemistry That Led to Life on Earth". The New York Times. Retrieved 5 May 2015.
- ^ S2CID 12277593.
- S2CID 29850349.
- PMID 1245233.
- PMID 21556207.
- PMID 25369814.
- .
- ^ Öberg, Karin (2020-04-10). The galactic recipe for a living planet. Retrieved 2024-12-24 – via www.ted.com.
- ^ doi:10.1086/180664.
- ISBN 978-0792345381.
- .
- doi:10.1086/317129.
- S2CID 122549795.
- ^ S2CID 118958200.
- S2CID 11335358.
- S2CID 9135663.
- S2CID 8016228.
- S2CID 14398456.
- ^ Zubritsky E, Neal-Jones N (11 August 2014). "Release 14-038 – NASA's 3-D Study of Comets Reveals Chemical Factory at Work". NASA. Retrieved 12 August 2014.
- S2CID 26277035.
- ^ "First detection of super-earth atmosphere". ESA/Hubble Information Centre. February 16, 2016.
- ^ Green, Jaime (5 December 2023). "What Is Life? - The answer matters in space exploration. But we still don't really know". The Atlantic. Archived from the original on 5 December 2023. Retrieved 15 December 2023.
- ^ Chang, Kenneth (14 December 2023). "Poison Gas Hints at Potential for Life on an Ocean Moon of Saturn - A researcher who has studied the icy world said "the prospects for the development of life are getting better and better on Enceladus."". The New York Times. Archived from the original on 14 December 2023. Retrieved 15 December 2023.
- from the original on 15 December 2023. Retrieved 15 December 2023.
- ISBN 3640233603.
- ^ Weapons of War - Poison Gas. firstworldwar.com
- ^ a b Environmental and Health Effects Archived 2012-11-30 at the Wayback Machine. Cyanidecode.org. Retrieved on 2012-06-02.
- ^ "Hydrogen Cyanide". Organisation for the Prohibition of Chemical Weapons. Retrieved 2009-01-14.
- ISBN 9780300067552.
- ^ "Blue Fume". Chemical Factory Draslovka a.s. Retrieved 2020-07-06.
- ^ "Uragan D2". 2015-07-17. Archived from the original on 2015-07-17. Retrieved 2022-10-19.
- ^ Binkov's Battlegrounds (April 27, 2022). "How would have WW2 gone if the US had not used nuclear bombs on Japan?". YouTube.Com. Retrieved June 23, 2022.
- ^ Pilkington, Ed (28 May 2021). "Arizona 'refurbishes' its gas chamber to prepare for executions, documents reveal". The Guardian. Retrieved 2022-06-14.
- ^ "The Poison Garden website". Thepoisongarden.co.uk. Archived from the original on 10 February 2020. Retrieved 18 October 2014.
- ^ "Documentation for Immediately Dangerous to Life or Health Concentrations (IDLHs) – 74908". NIOSH. 2 November 2018.
External links
- Institut national de recherche et de sécurité (1997). "Cyanure d'hydrogène et solutions aqueuses". Fiche toxicologique n° 4, Paris:INRS, 5pp. (PDF file, in French)
- International Chemical Safety Card 0492
- Hydrogen cyanide and cyanides (CICAD61)
- National Pollutant Inventory: Cyanide compounds fact sheet
- NIOSH Pocket Guide to Chemical Hazards
- Department of health review
- Density of Hydrogen Cyanide gas



