Dimethylheptylpyran
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 20–39 hours |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
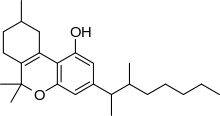
Dimethylheptylpyran (DMHP, 3-(1,2-dimethylheptyl)-Δ6a(10a)-THC, 1,2-dimethylheptyl-Δ3-THC, A-40824, or EA-2233) is a synthetic analog of THC, which was invented in 1949 during attempts to elucidate the structure of Δ9-THC, one of the active components of Cannabis.[2] DMHP is a pale yellow, viscous oil which is insoluble in water but dissolves in alcohol or non-polar solvents.
Effects
DMHP is similar in structure to THC, differing only in the position of one double bond, and the replacement of the 3-pentyl chain with a 3-(1,2-dimethylheptyl) chain.[3] It produces similar activity to THC, such as sedative effects, but is considerably more potent,[4] especially having much stronger analgesic and anticonvulsant effects than THC, although comparatively weaker psychological effects. It is thought to act as a CB1 agonist, in a similar manner to other cannabinoid derivatives.[5][6] While DMHP itself has been subject to relatively little study since the characterization of the cannabinoid receptors, the structural isomer 1,2-dimethylheptyl-Δ8-THC has been shown to be a highly potent cannabinoid agonist, and the activity of its enantiomers has been studied separately.[7]
Investigation as non-lethal incapacitating agent
DMHP and its O-
DMHP has three
DMHP is metabolized in a similar manner to THC, producing the active metabolite 11-hydroxy-DMHP, but the lipophilicity of DMHP is even higher than that of THC itself, giving it a long duration of action and an extended half-life in the body of between 20 and 39 hours, with the half-life of the 11-hydroxy-DMHP metabolite being longer than 48 hours.
DMHP and its esters produce
The acute toxicity of DMHP was found to be low in both human and animal studies, with the
Unsuitability for military application
The combination of strong incapacitating effects and a favorable safety margin led the Edgewood Arsenal team to conclude that DMHP and its derivatives, especially the O-acetyl ester of the most active isomer, EA-2233-2, were among the more promising non-lethal incapacitating agents to come out of their research program.
However, DMHP had the disadvantage of sometimes producing severe
Edgewood Arsenal and EA 2233
The fiscal budgeting and planning for Edgewood Arsenal (established in 1948) considered it to be primarily a defensive research facility. The US military, at the time, knew that the USSR was spending 10 times more than the USA on chemical weapons development. Edgewood initially enjoyed a mandate[clarification needed] and lack of oversight. Edgewood Arsenal Chemical Corps was tasked with ensuring that America was prepared with adequate counter-tactics if needed and that it could mount its own psychochemical retaliatory strike against the USSR if necessary. Edgewood performed analysis and submitted data to military commanders who could then choose to incorporate that into their strategy. In practice, however, it was mostly to make the strategists aware of special weapons and tactics that the enemy could instead deploy.
The Edgewood Laboratory was originally founded in 1948. The original
Multiple newspapers criticized the EA 2233 experiments. Since the 1930s, cannabis and cannabinoids had consistently been seen by the public as dangerous and addictive drugs. DMHP was compared to BZ, a non-cannabis chemical that was cited to be useless among military planners, and was only tested once in a hastily constructed operation called "Project Dork" (part of Project 112).[12]
Isomerism

| 7 double bond isomers of dimethylheptylpyran and their 120 stereoisomers | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Dibenzopyran numbering | Monoterpenoid numbering | Additional chiral centers on side chain | Number of stereoisomers | Natural occurrence | Convention on Psychotropic Substances Schedule | ||||
| Short name | Chiral centers in dibenzopyran backbone | Full name | Short name | Chiral centers in dibenzopyran backbone | 1,2-dimethylheptyl numbering | 3-methyloctan-2-yl numbering | |||
| Δ6a(7)-DMHP | 9 and 10a | 3-(1,2-dimethylheptyl)-8,9,10,10a-tetrahydro-6,6,9-trimethyl-6H-dibenzo[b,d]pyran-1-ol | Δ4-DMHP | 1 and 3 | 1 and 2 | 2 and 3 | 16 | No | unscheduled |
| Δ7-DMHP | 6a, 9 and 10a | 3-(1,2-dimethylheptyl)-6a,9,10,10a-tetrahydro-6,6,9-trimethyl-6H-dibenzo[b,d]pyran-1-ol | Δ5-DMHP | 1, 3 and 4 | 1 and 2 | 2 and 3 | 32 | No | unscheduled |
| Δ8-DMHP | 6a and 10a | 3-(1,2-dimethylheptyl)-6a,7,10,10a-tetrahydro-6,6,9-trimethyl-6H-dibenzo[b,d]pyran-1-ol | Δ6-DMHP | 3 and 4 | 1 and 2 | 2 and 3 | 16 | No | unscheduled |
| Δ9,11-DMHP | 6a and 10a | 3-(1,2-dimethylheptyl)-6a,7,8,9,10,10a-hexahydro-6,6-dimethyl-9-methylene-6H-dibenzo[b,d]pyran-1-ol | Δ1(7)-DMHP | 3 and 4 | 1 and 2 | 2 and 3 | 16 | No | unscheduled |
| Δ9-DMHP | 6a and 10a | 3-(1,2-dimethylheptyl)-6a,7,8,10a-tetrahydro-6,6,9-trimethyl-6H-dibenzo[b,d]pyran-1-ol | Δ1-DMHP | 3 and 4 | 1 and 2 | 2 and 3 | 16 | No | unscheduled |
| Δ10-DMHP | 6a and 9 | 3-(1,2-dimethylheptyl)-6a,7,8,9-tetrahydro-6,6,9-trimethyl-6H-dibenzo[b,d]pyran-1-ol | Δ2-DMHP | 1 and 4 | 1 and 2 | 2 and 3 | 16 | No | unscheduled |
| Δ6a(10a)-DMHP | 9 | 3-(1,2-dimethylheptyl)-7,8,9,10-tetrahydro-6,6,9-trimethyl-6H-dibenzo[b,d]pyran-1-ol | Δ3-DMHP | 1 | 1 and 2 | 2 and 3 | 8 | No | Schedule I |
Note that 6H-dibenzo[b,d]pyran-1-ol is the same as 6H-benzo[c]chromen-1-ol.
See also
- Tinabinol
- Nabitan
- Menabitan
- A-40174
- A-41988
- Δ-3-Tetrahydrocannabinol
- Tetrahydrocannabinol
- Tetrahydrocannabiphorol
- THCP-O-acetate
- Synthetic cannabinoids
- Effects of cannabis
References
- ^ Anvisa (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.
- .
- ISBN 978-0-471-05460-3.
- S2CID 35663464.
- PMID 817021.
- S2CID 38974868.
- .
- ISBN 978-0-309-07772-9.
- ^ ISBN 978-1-4243-0080-8.
- ISBN 978-0-309-07772-9.
- ISBN 978-1-4243-0080-8.
- ^ Khatchadourian R (12 December 2012). "War of the Mind". The New Yorker. Retrieved 2021-05-08.
