Isopropyl alcohol (data page)
This page provides supplementary chemical data on
isopropanol
.
Material Safety Data Sheet
The handling of this chemical may incur notable safety precautions. It is highly recommend that you seek the Material Safety Datasheet (
MSDS) for this chemical from a reliable source such as eChemPortal
, and follow its directions.
Structure and properties
| Structure and properties | |
|---|---|
Index of refraction , nD
|
1.3776 at 20°C |
| Abbe number | ? |
Dielectric constant , εr
|
18.23 ε0 at 25 °C |
Bond strength
|
? |
| Bond length | ? |
Bond angle
|
? |
| Magnetic susceptibility | ? |
| Surface tension | 21.7 dyn/cm at 20°C |
| Viscosity[1] | 4.5646 mPa·s at 0°C 2.3703 mPa·s at 20°C 1.3311 mPa·s at 40°C |
Thermodynamic properties
| Phase behavior | |
|---|---|
| Triple point | 184.9 K (−88.2 °C), ? Pa |
Critical point
|
508.7 K (235.6 °C), 5370 kPa |
Std enthalpy change , ΔfusHof fusion |
5.28 kJ/mol |
Std entropy change , ΔfusSof fusion |
28.6 J/(mol·K) |
Std enthalpy change , ΔvapHof vaporization |
44.0 kJ/mol |
Std entropy change , ΔvapSof vaporization |
124 J/(mol·K) |
| Solid properties | |
Std enthalpy change , ΔfHof formation |
? kJ/mol |
| Standard molar entropy, S |
? J/(mol K) |
| Heat capacity, cp | 0.212 J/(mol K) at −200°C |
| Liquid properties | |
Std enthalpy change , ΔfHof formation |
−318.2 kJ/mol |
| Standard molar entropy, S |
180 J/(mol K) |
| Heat capacity, cp | 2.68 J/(gK) at 20°C-25°C |
| Gas properties | |
Std enthalpy change , ΔfHof formation |
−261.1 kJ/mol |
| Standard molar entropy, S |
333 J/(mol K) |
| Heat capacity, cp | 1.54 J/(gK) at 25°C |
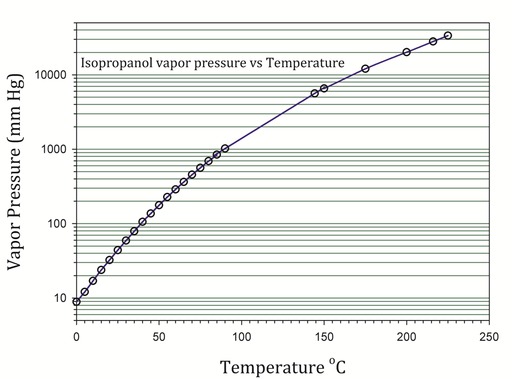
Vapor pressure of Iso-propyl Alcohol
| P in mm Hg | 1 | 10 | 40 | 100 | 400 | 760 | 1520 | 3800 | 7600 | 15200 | 30400 | 45600 | |
| T in °C | −26.1 | 2.4 | 23.8 | 39.5 | 67.8 | 82.5 | 101.3 | 130.2 | 155.7 | 186.0 | 220.2 | — | |
Table data obtained from CRC Handbook of Chemistry and Physics 44th ed.
Distillation data
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
See also
Spectral data
UV-Vis
| |
|---|---|
λmax
|
205 nm |
Extinction coefficient , ε
|
100.2 m2/mol |
| IR | |
| Major absorption bands | 3334, 2970, 1466, 1378, 1160, 1128, 951, 817, 639 cm−1 |
NMR
| |
Proton NMR
|
|
Carbon-13 NMR
|
|
| Other NMR data | |
MS
| |
| Masses of main fragments |
m/z (% of relative intensity): 45 (100), 43 (19.1), 27 (16.8), 29 (12.5), 19 (9.9), 15 (9.5), 41 (8.2), 31 (6.8), 39 (6.6), 42 (4.4) |
This box:
- Except where noted otherwise, data relate to Standard temperature and pressure.
- Reliability of data general note.
References
- ^ Lange's Handbook of Chemistry, 10th ed. pp 1669–1674
- .
- .
- ^ a b c "Binary Vapor-Liquid Equilibrium Data" (Queriable database). Chemical Engineering Research Information Center. Retrieved 5 May 2007.
