Acetone
| |||
| |||

| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Acetone[7]
| |||
| Preferred IUPAC name
Propan-2-one[8] | |||
| Systematic IUPAC name
2-Propanone | |||
| Other names | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 3DMet | |||
| 635680 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.000.602 | ||
| EC Number |
| ||
| 1466 | |||
| KEGG | |||
| MeSH | Acetone | ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1090 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H6O | |||
| Molar mass | 58.080 g·mol−1 | ||
| Appearance | Colourless liquid | ||
| Odor | Pungent, fruity[9] | ||
| Density | 0.7845 g/cm3 (25 °C)[10] | ||
| Melting point | −94.9 °C (−138.8 °F; 178.2 K)[10] | ||
| Boiling point | 56.08 °C (132.94 °F; 329.23 K)[10] | ||
| Miscible[10] | |||
| Solubility | Miscible in benzene, diethyl ether, methanol, chloroform, ethanol[10] | ||
| log P | −0.24[11] | ||
| Vapor pressure |
| ||
| Acidity (pKa) | |||
| −33.8·10−6 cm3/mol[14] | |||
Thermal conductivity
|
0.161 W/(m·K) (25 °C)[15] | ||
Refractive index (nD)
|
1.3588 (20 °C)[10] | ||
| Viscosity | 0.306 mPa·s (25 °C)[16] | ||
| Structure | |||
Trigonal planar at C2
| |||
| Dihedral at C2 | |||
| 2.88 D[17] | |||
| Thermochemistry[18] | |||
Heat capacity (C)
|
126.3 J/(mol·K) | ||
Std molar
entropy (S⦵298) |
199.8 J/(mol·K) | ||
Std enthalpy of (ΔfH⦵298)formation |
−248.4 kJ/mol | ||
Std enthalpy of (ΔcH⦵298)combustion |
−1.79 MJ/mol | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Highly flammable | ||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H225, H302, H319, H336, H373 | |||
| P210, P235, P260, P305+P351+P338 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | −20 °C (−4 °F; 253 K)[19] | ||
| 465[19] °C (869 °F; 738 K) | |||
Explosive limits
|
2.5–12.8%[19] | ||
Threshold limit value (TLV)
|
250 ppm[20] (STEL), 500 ppm[20] (C) | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
| ||
LC50 (median concentration)
|
20,702 ppm (rat, 8 h)[21] | ||
LCLo (lowest published)
|
45,455 ppm (mouse, 1 h)[21] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
1000 ppm (2400 mg/m3)[3] | ||
REL (Recommended)
|
TWA 250 ppm (590 mg/m3)[3] | ||
IDLH (Immediate danger) |
2500 ppm[3] | ||
| Related compounds | |||
Related compounds
|
|||
| Supplementary data page | |||
| Acetone (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Acetone (2-propanone or dimethyl ketone) is an
Acetone is
Acetone is produced and disposed of in the human body through normal metabolic processes. It is normally present in blood and urine. People with
Name
From the 17th century and before modern developments in
Prior to the "acetone" name given by
which were first synthesised from acetone.Unlike many compounds with the acet- prefix having a 2-carbon chain, acetone has a 3-carbon chain which has caused confusion since there cannot be a ketone with 2 carbons. The prefix refers to acetone's relation to vinegar (acetum in Latin, also the source of the words "acid" and "acetic"), rather than its chemical structure.[28]
History
Acetone was first produced by Andreas Libavius in 1606 by distillation of lead(II) acetate.[29][30]
In 1832, French chemist
Production
In 2010, the worldwide production capacity for acetone was estimated at 6.7 million tonnes per year.
Current method
Acetone is produced directly or indirectly from
Other processes involve the direct oxidation of propylene (
Older methods
Previously, acetone was produced by the dry distillation of acetates, for example calcium acetate in ketonic decarboxylation.
After that time, during
Chemical properties
The
Like most ketones, acetone exhibits the
In the presence of suitable
Acetone is a weak Lewis base that forms adducts with soft acids like I2 and hard acids like phenol. Acetone also forms complexes with divalent metals.[47][48]
Polymerisation
One might expect acetone to also form polymers and (possibly cyclic) oligomers of two types. In one type, units could be acetone molecules linked by ether bridges −O− derived from opening of the double bond, to give a polyketal-like (PKA) chain [−O−C(CH3)2−]n. The other type could be obtained through repeated aldol condensation, with one molecule of water removed at each step, yielding a poly(methylacetylene) (PMA) chain [−CH=C(CH3)−]n.[49]
The conversion of acetone to a polyketal (PKA) would be analogous to the formation of
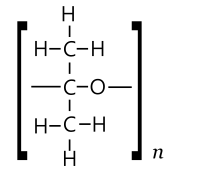
The PMA type polymers of acetone would be equivalent to the product of polymerisation of propyne, except for a keto end group.[49]
Natural occurrence
Humans exhale several milligrams of acetone per day. It arises from decarboxylation of
Metabolism
Acetone can then be metabolized either by
Uses
Industrial
About a third of the world's acetone is used as a solvent, and a quarter is consumed as acetone cyanohydrin, a precursor to methyl methacrylate.[23]
Solvent
Acetone is a good solvent for many plastics and some synthetic fibers. It is used for thinning
Although itself
Acetone is used as a solvent by the pharmaceutical industry and as a denaturant in denatured alcohol.[60] Acetone is also present as an
Chemical intermediate
Acetone is used to synthesize methyl methacrylate. It begins with the initial conversion of acetone to acetone cyanohydrin via reaction with hydrogen cyanide (HCN):
In a subsequent step, the nitrile is hydrolyzed to the unsaturated amide, which is esterified:
The third major use of acetone (about 20%)
Many millions of kilograms of acetone are consumed in the production of the solvents methyl isobutyl alcohol and methyl isobutyl ketone. These products arise via an initial aldol condensation to give diacetone alcohol.[24]
Condensation with acetylene gives 2-methylbut-3-yn-2-ol, precursor to synthetic terpenes and terpenoids.[62]
Laboratory
Chemistry
A variety of
Acetone freezes well below −78 °C. An acetone/dry ice mixture cools many a low-temperature reactions.[65]
Physics
Under ultraviolet light, acetone fluoresces. Fluid flow experiments use its vapor as a
Biology
In
Acetone also removes certain stains from microscope slides.[70]
Medical
Dermatologists use acetone with alcohol for acne treatments to chemically peel dry skin. Common agents used today for chemical peeling are salicylic acid, glycolic acid, azelaic acid, 30% salicylic acid in ethanol, and trichloroacetic acid (TCA). Prior to chemexfoliation, the skin is cleaned and excess fat removed in a process called defatting. Acetone, hexachlorophene, or a combination of these agents was used in this process.[71]
Acetone has been shown to have anticonvulsant effects in animal models of epilepsy, in the absence of toxicity, when administered in millimolar concentrations.[72] It has been hypothesized that the high-fat low-carbohydrate ketogenic diet used clinically to control drug-resistant epilepsy in children works by elevating acetone in the brain.[72] Because of their higher energy requirements, children have higher acetone production than most adults – and the younger the child, the higher the expected production. This indicates that children are not uniquely susceptible to acetone exposure. External exposures are small compared to the exposures associated with the ketogenic diet.[73]
Domestic and other niche uses
Make-up artists use acetone to remove skin adhesive from the netting of wigs and mustaches by immersing the item in an acetone bath, then removing the softened glue residue with a stiff brush.[74]
Acetone is often used for vapor polishing of printing artifacts on 3D-printed models printed with ABS plastic. The technique, called acetone vapor bath smoothing, involves placing the printed part in a sealed chamber containing a small amount of acetone, and heating to around 80 degrees Celsius for ten minutes. This creates a vapor of acetone in the container. The acetone condenses evenly all over the part, causing the surface to soften and liquefy. Surface tension then smooths the semi-liquid plastic. When the part is removed from the chamber, the acetone component evaporates leaving a glassy-smooth part free of striation, patterning, and visible layer edges, common features in untreated 3D printed parts.[75]
Acetone efficiently removes felt-tipped pen marks from glass and metals.
Safety
Acetone's most hazardous property is its extreme flammability. In small amounts, acetone burns with a
Acetone should be stored away from strong oxidizers, such as concentrated
Toxicity
Acetone occurs naturally as part of certain metabolic processes in the human body, and has been studied extensively and is believed to exhibit only slight toxicity in normal use. There is no strong evidence of chronic health effects if basic precautions are followed.[81] It is generally recognized to have low acute and chronic toxicity if ingested and/or inhaled.[82] Acetone is not currently regarded as a carcinogen, a mutagen, or a concern for chronic neurotoxicity effects.[76]
Acetone can be found as an ingredient in a variety of consumer products ranging from cosmetics to processed and unprocessed foods. Acetone has been rated as a generally recognized as safe (GRAS) substance when present in drinks, baked foods, desserts, and preserves at concentrations ranging from 5 to 8 mg/L.[82]
Acetone is however an irritant, causing mild skin and moderate-to-severe eye irritation. At high vapor concentrations, it may depress the central nervous system like many other solvents.[83] Acute toxicity for mice by ingestion (LD50) is 3 g/kg, and by inhalation (LC50) is 44 g/m3 over 4 hours.[84]
Environmental effects
Although acetone occurs naturally in the environment in plants, trees, volcanic gases, forest fires, and as a product of the breakdown of body fat,
EPA classification
In 1995, the United States Environmental Protection Agency (EPA) removed acetone from the list of volatile organic compounds. The companies requesting the removal argued that it would "contribute to the achievement of several important environmental goals and would support EPA's pollution prevention efforts", and that acetone could be used as a substitute for several compounds that are listed as hazardous air pollutants (HAP) under section 112 of the Clean Air Act.[87] In making its decision EPA conducted an extensive review of the available toxicity data on acetone, which was continued through the 2000s. It found that the evaluable "data are inadequate for an assessment of the human carcinogenic potential of acetone".[9]
Extraterrestrial occurrence
On 30 July 2015, scientists reported that upon the first touchdown of the Philae lander on comet 67P's surface, measurements by the COSAC and Ptolemy instruments revealed sixteen organic compounds, four of which were seen for the first time on a comet, including acetamide, acetone, methyl isocyanate, and propionaldehyde.[88][89][90]
References
- The Royal Society of Chemistry.(subscription required)
- ^ a b c d Acetone in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD)
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0004". National Institute for Occupational Safety and Health (NIOSH).
- ISBN 978-0-444-51994-8.
- ISBN 978-0-313-08057-9.
- ^ a b c Mel Gorman, History of acetone (1600-1850), 1962
- ^ ChemSpider lists 'acetone' as a valid, expert-verified name for what would systematically be called 'propan-2-one'.
- ISBN 978-0-85404-182-4.
- ^ a b Toxicological Profile for Acetone. U.S. Environmental Protection Agency June 2022 p. 7
- ^ a b c d e f Haynes, p. 3.4
- ^ Haynes, p. 5.173
- .
- S2CID 26624076.
- ^ Haynes, p. 3.576
- ^ Haynes, p. 6.254
- ^ Haynes, p. 6.243
- ^ Haynes, p. 9.60
- ^ Haynes, pp. 5.3, 5.67
- ^ a b c d Haynes, p. 15.13
- ^ a b Haynes, p. 16.34
- ^ a b c "Acetone". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- .
- ^ a b c Acetone, World Petrochemicals report, January 2010
- ^ a b c d e f Stylianos Sifniades, Alan B. Levy, "Acetone" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005.
- ^ "Update: U.S. EPA Exempt Volatile Organic Compounds". American Coatings Association. 2018-01-30. Archived from the original on 2021-02-08. Retrieved 2019-03-20.
- S2CID 26629499.
- ^ C. Reichenbach (1834) "Ueber Mesit (Essiggeist) und Holzgeist" (On mesit (spirit of vinegar) and wood spirits), Annalen der Pharmacie, vol. 10, no. 3, pages 298–314.
- ^ a b "Acetone". 28 September 2018.
- ^ Libavius, Andreas (1606). Alchymia (in Latin). Frankfurt, Germany: printed by Joannes Saurius, at the expense of Peter Kopff. p. 123.
- ^ "Aceton". Chemgapedia.
- ^ Dumas, J. (1832) "Sur l'esprit pyro-acétique" (On pyro-acetic spirit), Annales de Chimie et de Physique, 2nd series, 49 : 208–210.
- ^ Liebig, Justus (1832) "Sur les combinaisons produites par l'action du gas oléfiant et l'esprit acétique" (On compounds produced by the action of ethylene and acetic spirit), Annales de Chimie et de Physique, 2nd series, 49 : 146–204 (especially 193–204).
- ^ Bussy, Antoine (1833) "De quelques Produits nouveaux obtenus par l'action des Alcalis sur les Corps gras à une haute température" (On some new products obtained by the action of alkalies on fatty substances at a high temperature), Annales de Chimie et de Physique, 2nd series, 53 : 398–412; see footnote on pp. 408–409.
- ^ Williamson, A. W. (1852) "On Etherification," Journal of the Chemical Society, 4 : 229–239; (especially pp. 237–239).
- ^ Gerhardt, Charles (1853) "Researches sur les acids organiques anhydres" (Research on anhydrous organic acids), Annales de Chimie et de Physique, 3rd series, 37 : 285–342; see p. 339.
- ^ Kekulé, Auguste (1865) "Sur la constitution des substances aromatiques," Bulletin de la Société chimique de Paris, 1 : 98–110; (especially p. 110).
- ^ Kekulé, Auguste (1866) "Untersuchungen über aromatischen Verbindungen" (Investigations into aromatic compounds), Annalen der Chemie und Pharmacie, 137 : 129–196; (especially pp. 143–144).
- ^ Loschmidt, J. (1861) Chemische Studien Vienna, Austria-Hungary: Carl Gerold's Sohn.
- ^ Chaim Weizmann chemistryexplained.com
- ^ a b Greiner, Camara; Funada, C (June 2010). "CEH Marketing Research Report: ACETONE". Chemical Economics Handbook. SRI consulting. Retrieved 2 September 2016.(subscription required)
- ^ "Acetone Uses and Market Data". ICIS.com. October 2010. Archived from the original on 2009-05-15. Retrieved 2011-03-21.
- ^ Acetone (US Gulf) Price Report – Chemical pricing information Archived 2013-05-16 at the Wayback Machine. ICIS Pricing, Retrieved on 2012-11-26
- ISBN 3-527-30289-1.
- ^ Haynes, p. 15.49
- .
- OCLC 67767141.
- .
- PMID 16954630.
- ^ .
- ^ Kargin, V. A.; Kabanov, V. A.; Zubov, V. P.; Papisov, I. M. (1960). "Polymerisation of acetone". Doklady Akademii Nauk SSSR. 134 (5): 1098–1099.
- .
- ^ OCLC 61503700.
- S2CID 40583110.
- ^ Glew, Robert H (2010). "You Can Get There From Here: Acetone, Anionic Ketones and Even-Carbon Fatty Acids can Provide Substrates for Gluconeogenesis". Nig. J. Physiol. Sci. 25: 2–4. Archived from the original on 2013-09-26. Retrieved 2013-09-01.
- S2CID 37769342.
- PMID 4553872.
- OCLC 1104227601.)
{{cite book}}: CS1 maint: location missing publisher (link - ^ Mine Safety and Health Administration (MSHA) – Safety Hazard Information – Special Hazards of Acetylene Archived 2016-01-22 at the Wayback Machine. Msha.gov. Retrieved on 2012-11-26.
- ^ History – Acetylene dissolved in acetone Archived 2015-09-15 at the Wayback Machine. Aga.com, Retrieved on 2012-11-26
- ISBN 978-0-8247-8210-8.
- ^ Inactive Ingredient Search for Approved Drug Products, FDA/Center for Drug Evaluation and Research
- OCLC 53307689.
- ^ "Cleaning Glassware" (PDF). Wesleyan University. September 2009. Retrieved July 7, 2016.
- ^ What is an Azeotrope?. Solvent—recycling.com. Retrieved on 2012-11-26.
- ISBN 9780935702767.)
{{cite book}}: CS1 maint: location missing publisher (link - S2CID 121060565.
- ^ PMID 20000691.
- S2CID 195684316.
- S2CID 205030844.
- PMID 86548.
- OCLC 663098001.
- ^ S2CID 3213318.
- ^ American Chemistry Council Acetone Panel (September 10, 2003). "Acetone (CAS No. 67-64-1) VCCEP Submission" (PDF). pp. 6, 9. Retrieved 2018-04-14.
- OCLC 776632427.
- ^ "Quality Finish 3D Prints with Acetone" instructables.com
- ^ a b c "Acetone MSDS". hazard.com. 1998-04-21. Archived from the original on 2012-07-09. Retrieved 2012-11-26.
{{cite web}}: CS1 maint: unfit URL (link) - )
- ^ Haynes, p. 16.3
- ^ Haynes, p. 16.5
- )
- ^ Basic Information on Acetone. Ccohs.ca (1999-02-19). Retrieved on 2012-11-26.
- ^ a b "SIDS Initial Assessment Report: Acetone" (PDF). Environmental Protection Agency. Archived from the original (PDF) on 2014-03-09. Retrieved 2014-09-11.
- ^ "What are the potential health effects of acetone?". Canadian Centre for Occupational Health and Safety. Archived from the original on 2008-10-17. Retrieved 2008-10-21.
- ^ Safety (MSDS) data for propanone Archived 2018-03-16 at the Wayback Machine sciencelab.com/msds Retrieved on 2018-03-19
- ^ a b Acetone, Agency for Toxic Substances and Disease Registry ToxFAQs, 1995
- .
- ^ U.S. Environmental Protection Agency (June 16, 1995). "Air Quality: Revision to Definition of Volatile Organic Compounds—Exclusion of Acetone" (PDF). Federal Register. 60 (116): 31634–31637.
- ^ Jordans, Frank (30 July 2015). "Philae probe finds evidence that comets can be cosmic labs". The Washington Post. Associated Press. Archived from the original on 23 December 2018. Retrieved 30 July 2015.
- ^ "Science on the Surface of a Comet". European Space Agency. 30 July 2015. Retrieved 30 July 2015.
- PMID 26228139.
Common sources
- Haynes, William M., ed. (2016). ISBN 9781498754293.
Further reading
- International Chemical Safety Card 0087
- NIOSH Pocket Guide to Chemical Hazards
- Acetone Safety Data Sheet (SDS)
- Hazardous substances databank entry at the national library of medicine Archived 2018-12-04 at the Wayback Machine
- SIDS Initial Assessment Report for Acetone from the Organisation for Economic Co-operation and Development(OECD)
- Calculation of vapor pressure, liquid density, dynamic liquid viscosity, surface tension of acetone












