Staggered conformation
Appearance

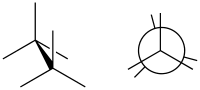
In
torsion angles to be 60°.[1] It is the opposite of an eclipsed conformation
, in which those substituents are as close to each other as possible.
Such a conformation exists in any open chain single chemical bond connecting two sp3-hybridised atoms, and is normally a conformational energy minimum. For some molecules such as those of n-butane, there can be special versions of staggered conformations called gauche and anti; see first Newman projection diagram in Conformational isomerism.
Staggered/eclipsed configurations also distinguish different crystalline structures of e.g. cubic/hexagonal boron nitride, and diamond/lonsdaleite.
See also
- Alkane stereochemistry
- Eclipsed conformation
References
- ISBN 978-0-471-01670-0.
