Eclipsed conformation
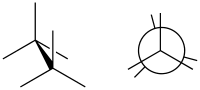
(image right in Newman projection)

In
In order to gain a deeper understanding of eclipsed conformations in organic chemistry, it is first important to understand how organic molecules are arranged around bonds, as well as how they move and rotate.
In the example of ethane, two methyl groups are connected with a carbon-carbon sigma bond, just as one might connect two Lego pieces through a single “stud” and “tube”. With this image in mind, if the methyl groups are rotated around the bond, they will remain connected; however, the shape will change. This leads to multiple possible three-dimensional arrangements, known as conformations, conformational isomers (conformers), or sometimes rotational isomers (rotamers).[2]
Organic chemistry
Conformations can be described by

In the case of butane
If the front is rotated 60° clockwise, the butane molecule is now in a staggered conformation (right). This conformation is more specifically referred to as the
The minima can be seen on the graph at 60, 180 and 300 degrees while the maxima can bee see at 0, 120, 240, and 360 degrees. The maxima represent the eclipsed conformations due to the dihedral angle of zero degrees.[4]
Structural applications
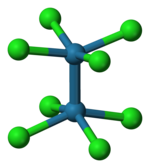
As established by X-ray crystallography, octachlorodimolybdate(II) anion ([Mo2Cl8]4-) has an eclipsed conformation. This sterically unfavorable geometry is given as evidence for a quadruple bond between the Mo centers.[5]
Experiments such as X-ray and electron diffraction analyses, nuclear magnetic resonance, microwave spectroscopies, and more have allowed researchers to determine which cycloalkane structures are the most stable based on the different possible conformations. Another method that was shown successful is molecular mechanics, a computational method that allows the total strain energies of different conformations to be found and analyzed. It was found that the most stable conformations had lower energies based on values of energy due to bond distances and bond angles.[6]
In many cases, isomers of alkanes with branched chains have lower boiling points than those that are unbranched, which has been shown through experimentation with isomers of C8H18. This is because of a combination of intermolecular forces and size that results from the branched chains. The more branches that an alkane has, the more extended its shape is; meanwhile, if it is less branched then it will have more intermolecular attractive forces that will need to be broken which is the cause of the increased boiling point for unbranched alkanes. In another case,
See also
References
- ISBN 978-0-471-01670-0.
- ^ Ashenhurst, James (February 28, 2020). "Staggered vs Eclipsed Conformations of Ethane". Mastering Organic Chemistry. Retrieved November 15, 2022.
- ^ "hydrocarbon - Three-dimensional structures | Britannica". www.britannica.com. Retrieved 2022-11-15.
- ^ "3.4.1. Newman Projections". Chemistry LibreTexts. 2015-06-16. Retrieved 2022-11-15.
- ISBN 978-0-470-13244-9.
- ^ a b "hydrocarbon - Three-Dimensional Structures". Britannica. Retrieved November 15, 2022.
