Wagner–Meerwein rearrangement
A Wagner–Meerwein rearrangement is a class of
aryl group migrates from one carbon to a neighboring carbon.[1]
Yegor Yegorovich Vagner; he had German origin and published in German journals as Georg Wagner; and Hans Meerwein
.
Several reviews have been published.[3][4][5][6][7]
The rearrangement was first discovered in

The story of the rearrangement reveals that many scientists were puzzled with this and related reactions and its close relationship to the discovery of
carbocations as intermediates.[9]
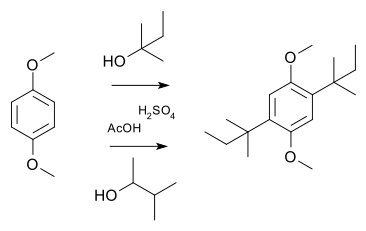
In a simple demonstration reaction of
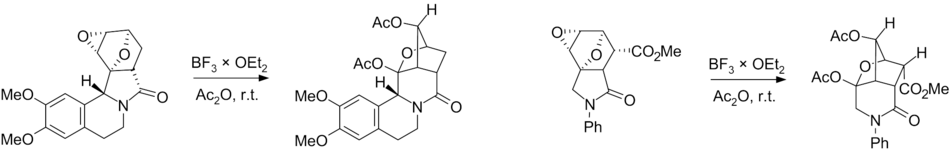
the latter via a hydride shift of the cationic intermediate:Currently, there are works relating to the use of skeletal rearrangement in the synthesis of bridged azaheterocycles. These data are summarized in [11]
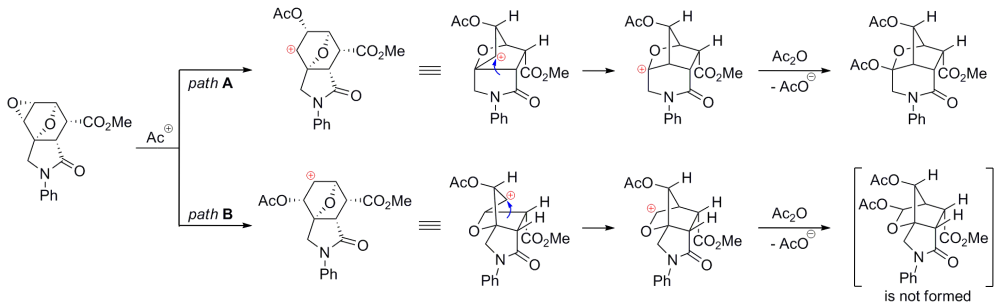
Plausible mechanisms of the Wagner–Meerwein rearrangement of diepoxyisoindoles:
The related Nametkin rearrangement, named after
Sergey Namyotkin, involves the rearrangement of methyl groups in certain terpenes. In some cases the reaction type is also called a retropinacol rearrangement (see pinacol rearrangement
).
References
- Vagner, Ye. Ye. (Wagner, G.) (1899). "In "Protokol zasedaniya Otdeleniya Khimii R. F. Khimicheskago Obshchestva. 9-go sentyabrya 1899 goda [Minutes of the meeting of the Chemistry Section of the Russian Physical-Chemical Society. 9th September 1899]"". J. Russ. Phys. Chem. Soc. [Zh. Russ. Fiz.-Khim. O-va.] 31: 680–684.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - .
- .
- .
- .
- ISBN 3-540-09309-5.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ISBN 978-0-08-052349-1.
- OCLC 642506595
- .
- .
- .