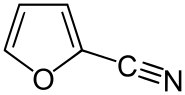
2-Furonitrile
| |
| Names | |
|---|---|
| Preferred IUPAC name
Furan-2-carbonitrile | |
| Other names
2-Cyanofuran; 2-Furancarbonitrile; 2-Furyl cyanide
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.009.581 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C5H3NO | |
| Molar mass | 93.085 g·mol−1 |
| Appearance | colorless (yellow if impure) |
| Density | 1.0650 @20 °C [1] |
| Boiling point | 147[2] °C (297 °F; 420 K) |
| Hazards | |
| Flash point | 35 °C; 95 °F; 308 K |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
2-Furonitrile is a colorless derivative of furan possessing a nitrile group.
Synthesis
Industrial synthesis is based on the vapor phase ammoxidation of furfural with ammonia over bismuth molybdate catalyst at 440-480 °C.[3]
Numerous laboratory methods also exist; for the instance oxidative dehydration of furfural with
Applications
2-Furonitrile currently has no major applications but it is used as an intermediate in pharmaceutical and fine chemical synthesis. It has been suggested as a potential sweetening agent, as it has about 30 times the sweetening power of sucrose.[10]
References
- ^ P. A. Pavlov; Kul'nevich, V. G. (1986). "Synthesis of 5-substituted furannitriles and their reaction with hydrazine". Khimiya Geterotsiklicheskikh Soedinenii. 2: 181–186.
- S2CID 97316774.
- ^ Thomas J. Jennings, "Process for preparing furonitrile", US Patent 3,260,731 (1966)
- .
- S2CID 98593006.
- .
- .
- ISSN 0040-4039.
- .
- ^ Thomas J. Jennings, "Process for preparing furonitrile", US Patent 3,260,731 (1966)
